Search
- Page Path
- HOME > Search
Research Article
- [English]
- Effect of Compositional Trade-off Between Cr and Mo on the Corrosion Resistance of Additively Manufactured Co-Cr-Fe-Ni-Mo High-Entropy Alloys
- Jeongmin Lee, Yeonghwan Song, Jae Hyuk Lee, Sung-Jae Jo, Minho Shin, Hyunbin Lim, Soon-Jik Hong, Soo-Hyun Joo
- J Powder Mater. 2026;33(2):137-144. Published online April 30, 2026
- DOI: https://doi.org/10.4150/jpm.2026.00087
- 656 View
- 9 Download
-
 Abstract
Abstract
 PDF
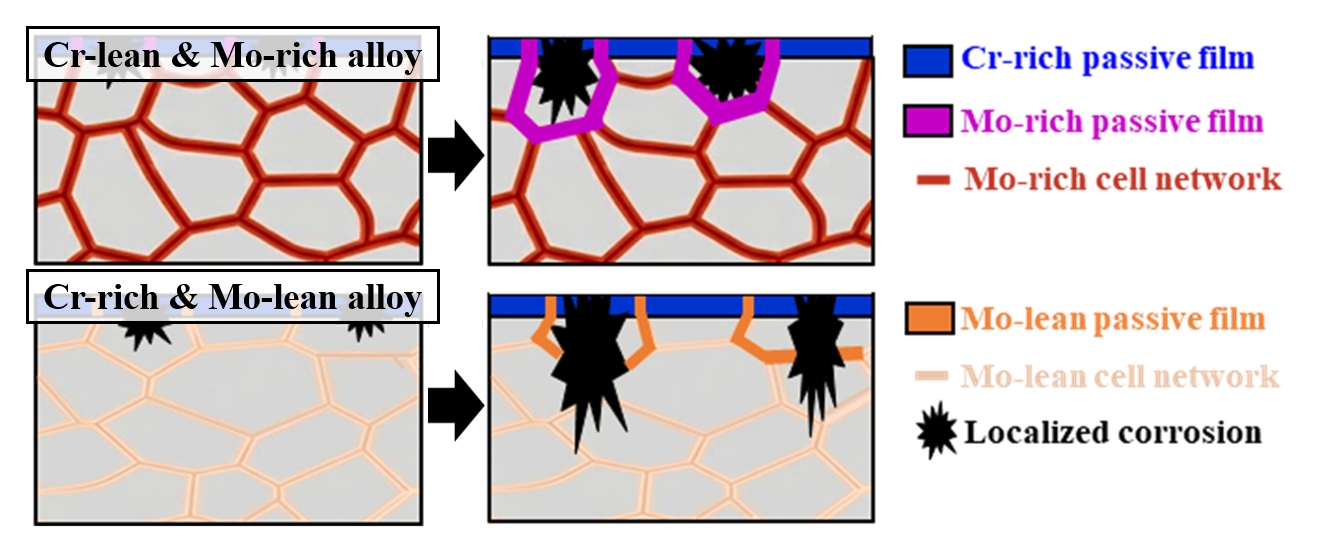
PDF - In this study, the corrosion behavior of Co-Cr-Fe-Ni-Mo high-entropy alloys additively manufactured via direct energy deposition was investigated according to the compositional trade-off between Cr and Mo elements. Two distinct alloy compositions were fabricated by adjusting the feeding rate of two powders with different chemical compositions through a dual nozzle. Electrochemical testing in a 3.5 wt% NaCl solution revealed that the Cr-rich and Mo-lean alloy exhibited inferior corrosion resistance compared to the Cr-lean and Mo-rich alloy. Specifically, the corrosion potential of the Cr-rich and Mo-lean alloy shifted negatively by approximately 200 mV compared to the Cr-lean and Mo-rich alloy, accompanied by an increase in corrosion current density and the pronounced initiation of localized pitting. This deterioration is attributed to a lack of passivation caused by the small amount of Mo in the Cr-rich and Mo-lean alloy. The passive film of the Cr-lean and Mo-rich alloy was more robust, characterized by a higher concentration of Mo, which effectively inhibited pit propagation through repassivation. These findings demonstrate that maintaining a critical Cr-Mo balance is more vital for the electrochemical stability of additively manufactured high-entropy alloys than unilateral Cr enrichment.
TOP
 KPMI
KPMI

 First
First Prev
Prev


