Search
- Page Path
- HOME > Search
- [Korean]
- Viologen-based All-in-one Electrochromic Devices with a Lateral Electrode Structure
- Hyun-Jeong Kim, Sang-Rae Lee, Jin-Hee Choi, Yoon-Chae Nah
- J Korean Powder Metall Inst. 2020;27(1):58-62. Published online February 1, 2020
- DOI: https://doi.org/10.4150/KPMI.2020.27.1.58
- 1,538 View
- 14 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
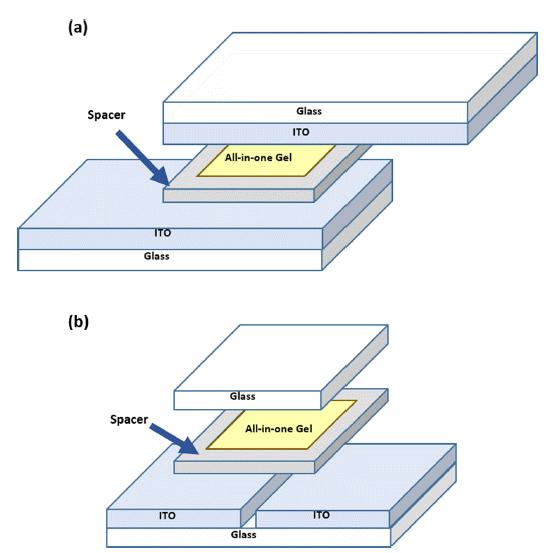
PDF Recently, electrochromic devices (ECDs) have gathered increasing attention owing to their high color contrast and memory effect, which make them highly applicable to smart windows, auto-dimming mirrors, sensors, etc. Traditional ECDs have a sandwich structure that contains an electrochromic layer between two ITO substrates. These sandwich-type devices are usually fabricated through the lamination of two electrodes and followed by the injection of a liquid electrolyte in the inner space. However, this process is sometimes complex and time consuming. In this study, we fabricated ECDs with a lateral electrode structure that uses only an ITO substrate and an all-in-one electrochromic gel, which is a mixture of electrolyte and electrochromic material. Furthermore, we investigated the EC properties of the lateral-type device by comparing it with a sandwich-type device. The lateral-type ECD shows strong blue absorption as the applied voltage increases and has a competitive coloration efficiency compared to the sandwich-type device.
-
Citations
Citations to this article as recorded by- Viologens and extended viologen derivatives with mono- and di-hexyl substituents for highly stable all-in-one ECDs and solar cell powered large-area ECDs
Fangyuan Sun, He Zhang, Cong Wang, Huan Ling, Jiayi Cai, Weilun Peng, Yanhong Tian, Fengyu Su, Yanqing Tian, Yan Jun Liu
Materials Chemistry and Physics.2023; 301: 127593. CrossRef
- Viologens and extended viologen derivatives with mono- and di-hexyl substituents for highly stable all-in-one ECDs and solar cell powered large-area ECDs
- [Korean]
- High-Contrast Electrochromism of Porous Tungsten Oxide Thin Films Prepared by Electrodeposition
- Sung-Hyeok Park, Ho-Jin Mo, Jae-Keun Lim, Sang-Gwon Kim, Jae-Hyo Choi, Seung-Hyun Lee, Se-Hwa Jang, Kyung-Ho Cha, Yoon-Chae Nah
- J Korean Powder Metall Inst. 2018;25(1):7-11. Published online February 1, 2018
- DOI: https://doi.org/10.4150/KPMI.2018.25.1.7
- 1,256 View
- 17 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
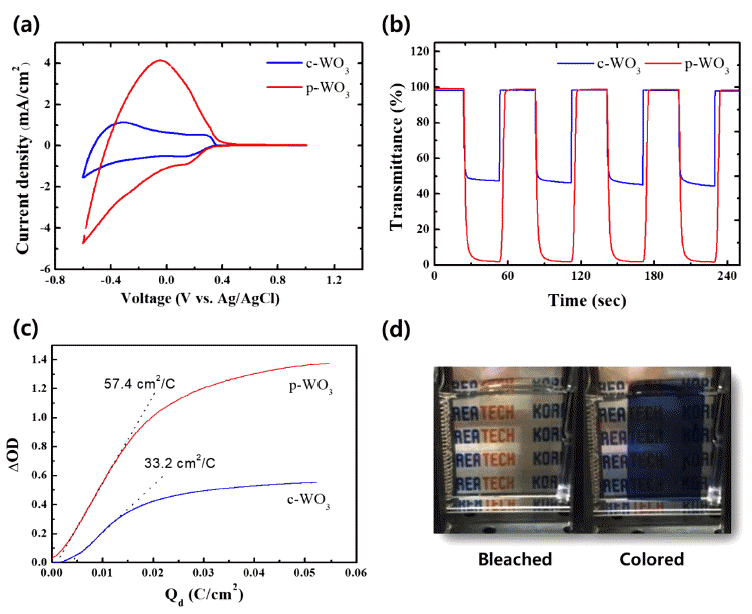
PDF In this study, we synthesize tungsten oxide thin films by electrodeposition and characterize their electrochromic properties. Depending on the deposition modes, compact and porous tungsten oxide films are fabricated on a transparent indium tin oxide (ITO) substrate. The morphology and crystal structure of the electrodeposited tungsten oxide thin films are investigated by scanning electron microscopy (SEM) and X-ray diffraction (XRD). X-ray photoelectron spectroscopy is employed to verify the chemical composition and the oxidation state of the films. Compared to the compact tungsten oxides, the porous films show superior electrochemical activities with higher reversibility during electrochemical reactions. Furthermore, they exhibit very high color contrast (97.0%) and switching speed (3.1 and 3.2 s). The outstanding electrochromic performances of the porous tungsten oxide thin films are mainly attributed to the porous structure, which facilitates ion intercalation/deintercalation during electrochemical reactions.
-
Citations
Citations to this article as recorded by- A fast-response electrochromic device based on a composite gel film comprising triphenylamine derivatives and WO3
Xuejian Zhang, Jinming Zeng, Zipeng Xu, Mimi Zhu, Ping Liu
New Journal of Chemistry.2021; 45(12): 5503. CrossRef
- A fast-response electrochromic device based on a composite gel film comprising triphenylamine derivatives and WO3
- [Korean]
- Effects of Heat Treatment and Viologen Incorporation on Electrochromic Properties of TiO2 Nanotubes
- Hyeongcheol Cha, Yoon-Chae Nah
- J Korean Powder Metall Inst. 2016;23(2):102-107. Published online April 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.2.102

- 1,091 View
- 5 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF We demonstrate the electrochromic properties of TiO2 nanotubes prepared by an anodization process and investigate the effects of heat treatment and viologen incorporation on them. The morphology and crystal structure of anodized TiO2 nanotubes are investigated by scanning electron microscopy and X-ray diffraction. As-formed TiO2 nanotubes have straight tubular layers with an amorphous structure. As the annealing temperature increases, the anodized TiO2 nanotubes are converted to the anatase and rutile phases with some cracks on the tube surface and irregular morphology. Electrochemical results reveal that amorphous TiO2 nanotubes annealed at 150°C have the largest oxidation/ reduction current, which leads to the best electrochromic performance during the coloring/bleaching process. Viologenanchored TiO2 nanotubes show superior electrochromic properties compared to pristine TiO2 nanotubes, which indicates that the incorporation of a viologen can be an effective way to enhance the electrochromic properties of TiO2 nanotubes.
-
Citations
Citations to this article as recorded by- Fabrication of patterned TiO2 nanotube layers utilizing a 3D printer platform and their electrochromic properties
Kwang-Mo Kang, Seok-Han Lee, Sang-Youn Kim, Yoon-Chae Nah
Electrochemistry Communications.2024; 169: 107833. CrossRef
- Fabrication of patterned TiO2 nanotube layers utilizing a 3D printer platform and their electrochromic properties
- [Korean]
- Fabrication of Photoelectrochromic Devices Composed of Anodized TiO2 and WO3 Nanostructures
- Sanghoon Lee, Hyeongcheol Cha, Yoon-Chae Nah
- J Korean Powder Metall Inst. 2015;22(5):326-330. Published online October 1, 2015
- DOI: https://doi.org/10.4150/KPMI.2015.22.5.326
- 830 View
- 4 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
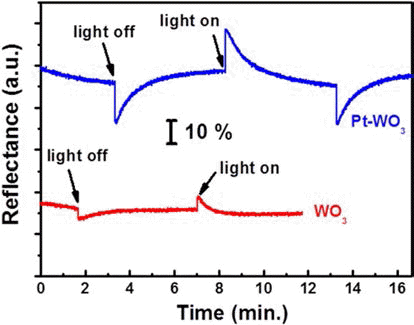
PDF In this study, we demonstrate the photoelectrochromic devices composed of TiO2 and WO3 nanostructures prepared by anodization method. The morphology and the crystal structure of anodized TiO2 nanotubes and WO3 nanoporous layers are investigated by SEM and XRD. To fabricate a transparent photoelectrode on FTO substrate, a TiO2 nanotube membrane, which has been detached from Ti substrate, is transferred to FTO substrate and annealed at 450°C for 1 hr. The photoelectrode of TiO2 nanotube and the counter electrode of WO3 nanoporous layer are assembled and the inner space is filled with a liquid electrolyte containing 0.5 M LiI and 5 mM I2 as a redox mediator. The properties of the photoelectrochromic devices is investigated and Pt-WO3 electrode system shows better electrochromic performance compared toWO3 electrode.
-
Citations
Citations to this article as recorded by- Synthesis and characterization of nitrogen-doped TiO2 coatings on reduced graphene oxide for enhancing the visible light photocatalytic activity
Yifan Zhang, Hye Mee Yang, Soo-Jin Park
Current Applied Physics.2018; 18(2): 163. CrossRef - Photocatalytic and Adsorption Properties of WO3 Nanorods Prepared by Hydrothermal Synthesis
Su-Yeol Yu, Chunghee Nam
Journal of Korean Powder Metallurgy Institute.2017; 24(6): 483. CrossRef - Synthesis and Photo Catalytic Activity of 10 wt%, 20 wt%Li-TiO2 Composite Powders
Hyeong-Chul Kim, Jae-Kil Han
Journal of Korean Powder Metallurgy Institute.2016; 23(1): 33. CrossRef
- Synthesis and characterization of nitrogen-doped TiO2 coatings on reduced graphene oxide for enhancing the visible light photocatalytic activity
- [English]
- Synthesis and Characterization of Tungsten Trioxide Films Prepared by a Sol-Gel Method for Electrochromic Applications
- Tae-Ho Kim, Yoon-Chae Nah
- J Korean Powder Metall Inst. 2015;22(5):309-314. Published online October 1, 2015
- DOI: https://doi.org/10.4150/KPMI.2015.22.5.309
- 1,218 View
- 5 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
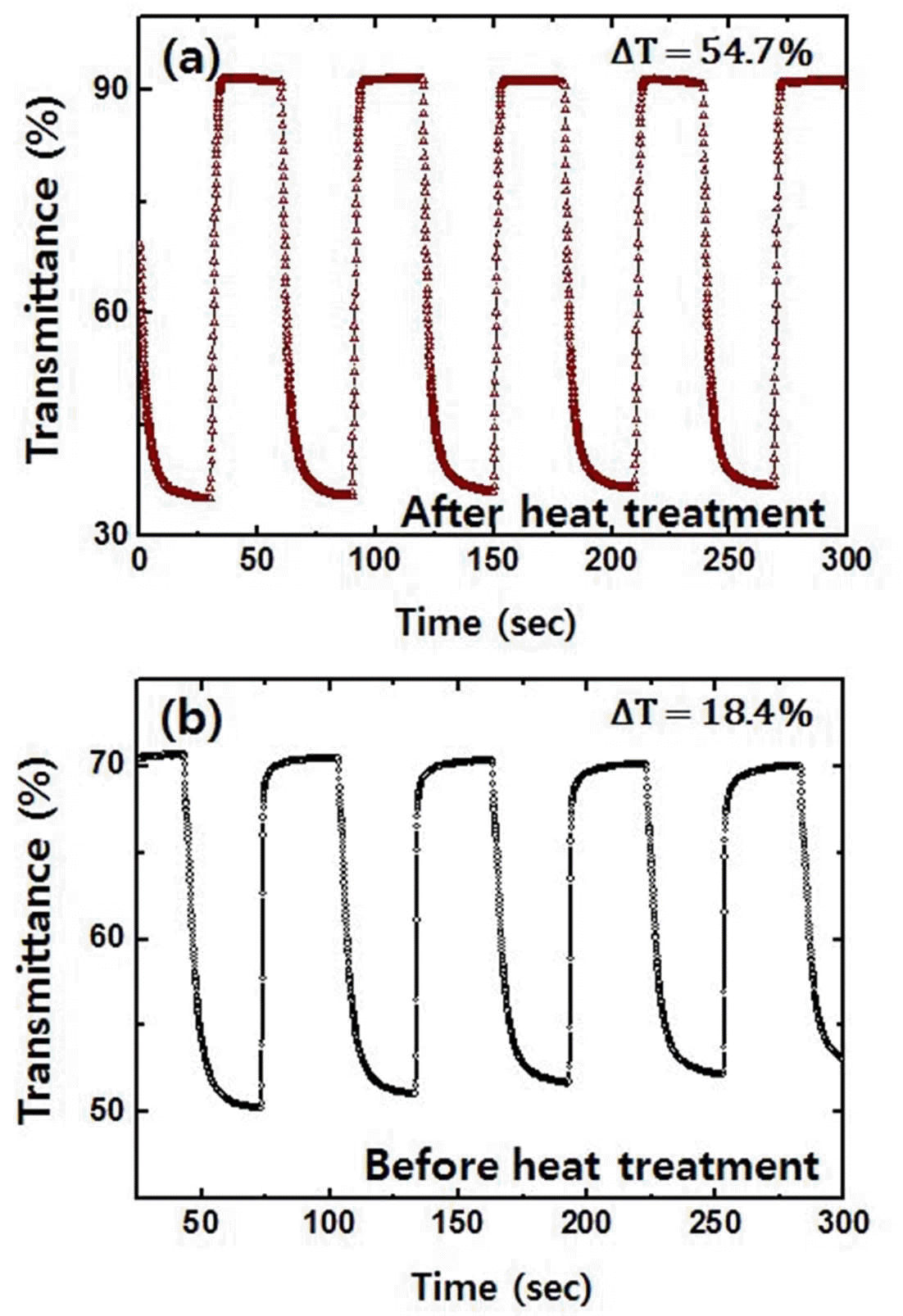
PDF Tungsten trioxide thin films are successfully synthesized by a sol-gel method using tungsten hexachloride as precursors. The structural, chemical, and optical properties of the prepared films are characterized by scanning electron microscopy, X-ray diffraction, X-ray photoelectron spectroscopy, and UV-Vis spectrophotometry. The electrochemical and electrochromic properties of the films before and after heat treatment are also investigated by cyclic voltammetry, chronoamperometry, and in situ transmittance measurement system. Compared to as-prepared films, heattreated tungsten trioxide thin films exhibit a higher electrochemical reversibility of 0.81 and superior coloration efficiency of 65.7 cm2/C, which implies that heat treatment at an appropriate temperature is a crucial process in a sol-gel method for having a better electrochromic performance.
-
Citations
Citations to this article as recorded by- Temperature-dependent electrochromic cycling performance of solution-processed WO3 films
Hoon Kang, Gyung Hyun Kim, Sungho Kang, Tae Hoon Park, Kwanchul Kim, Hwan Kyu Kim, Yekyung Kim
Nanoscale.2025; 17(35): 20135. CrossRef
- Temperature-dependent electrochromic cycling performance of solution-processed WO3 films
TOP
 KPMI
KPMI


 First
First Prev
Prev


