Search
- Page Path
- HOME > Search
- [Korean]
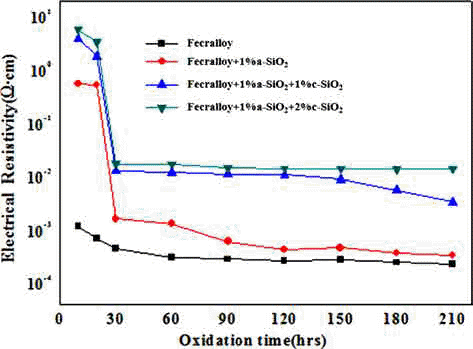
- The Effect of SiO2 addition on Oxidation and Electrical Resistance Stability at High-temperature of P/M Fecralloy Compact
- Jin-Woo Park, Jin-Uk Ok, Woo-young Jung, Dong-kyu Park, In-Shup Ahn
- J Korean Powder Metall Inst. 2017;24(4):292-297. Published online August 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.4.292
- 720 View
- 3 Download
-
 Abstract
Abstract
 PDF
PDF A metallic oxide layer of a heat-resistant element contributes to the high-temperature oxidation resistance by delaying the oxidation and has a positive effect on the increase in electrical resistivity. In this study, green compacts of Fecralloy powder mixed with amorphous and crystalline silica are oxidized at 950°C for up to 210 h in order to evaluate the effect of metal oxide on the oxidation and electrical resistivity. The weight change ratio increases as per a parabolic law, and the increase is larger than that observed for Fecralloy owing to the formation of Fe-Si, Fe-Cr composite oxide, and Al2O3 upon the addition of Si oxide. Si oxides promote the formation of Al2O3 and Cr oxide at the grain boundary, and obstruct neck formation and the growth of Fecralloy particles to ensure stable electrical resistivity.
- [Korean]
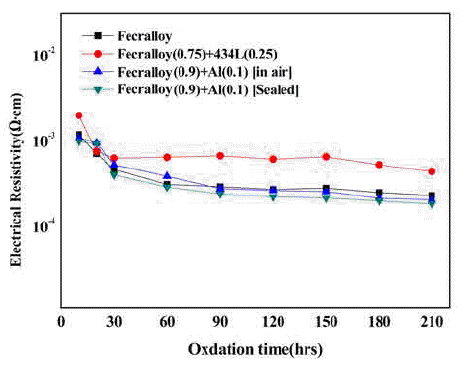
- A Study on the Development of Compactability and Electrical Resistivity for P/M Fecralloy
- Jin-Woo Parka, Byung-Hyun Ko, Woo-Young Jung, Dong-Kyu Park, In-Shup Ahn
- J Korean Powder Metall Inst. 2016;23(6):426-431. Published online December 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.6.426
- 662 View
- 2 Download
-
 Abstract
Abstract
 PDF
PDF The Fe-Cr-Al alloy system shows an excellent heat resistance because of the formation of an Al2O3 film on the metal surface in an oxidizing atmosphere at high temperatures up to 1400°C. The Fecralloy needs an additive that can act as a binder because of its bad compactability. In this study, the green compacts of STS434L and Al powder added to Fecralloy are oxidized at 950°C for up to 210 h. Fecralloy and Al is mixed by two types of ball milling. One is vented to air and the other was performed in a sealed jar. In the case of Al addition, there are no significant changes in the electrical resistance. Before the oxidation test, Al oxides are present in the Fecralloy surface, as determined from the energy dispersive spectroscopy results. The addition of Al improves the compactability because of an increased density, and the addition of STS434L increases the electrical resistivity by forming a composite oxide.
TOP
 KPMI
KPMI

 First
First Prev
Prev


