Search
- Page Path
- HOME > Search
- [Korean]
- A Study on Particle and Crystal Size Analysis of Lithium Lanthanum Titanate Powder Depending on Synthesis Methods (Sol-Gel & Solid-State reaction)
- Jeungjai Yun, Seung-Hwan Lee, So Hyun Baek, Yongbum Kwon, Yoseb Song, Bum Sung Kim, Bin Lee, Rhokyun Kwak, Da-Woon Jeong
- J Powder Mater. 2023;30(4):324-331. Published online August 1, 2023
- DOI: https://doi.org/10.4150/KPMI.2023.30.4.324
- 1,672 View
- 19 Download
-
 Abstract
Abstract
 PDF
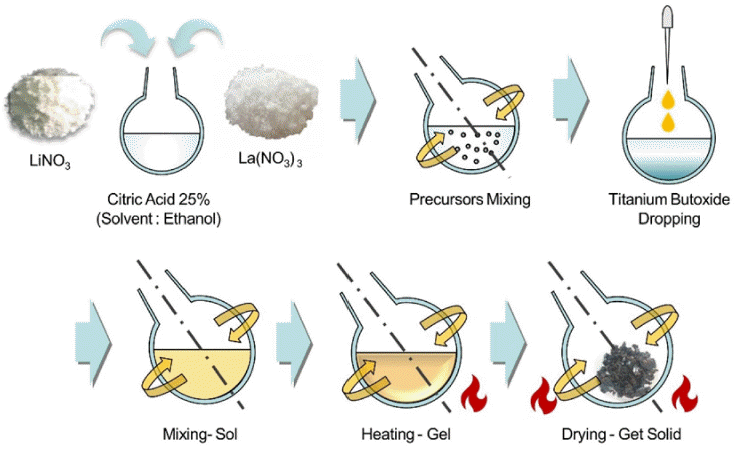
PDF Lithium (Li) is a key resource driving the rapid growth of the electric vehicle industry globally, with demand and prices continually on the rise. To address the limited reserves of major lithium sources such as rock and brine, research is underway on seawater Li extraction using electrodialysis and Li-ion selective membranes. Lithium lanthanum titanate (LLTO), an oxide solid electrolyte for all-solid-state batteries, is a promising Li-ion selective membrane. An important factor in enhancing its performance is employing the powder synthesis process. In this study, the LLTO powder is prepared using two synthesis methods: sol-gel reaction (SGR) and solid-state reaction (SSR). Additionally, the powder size and uniformity are compared, which are indices related to membrane performance. X-ray diffraction and scanning electron microscopy are employed for determining characterization, with crystallite size analysis through the full width at half maximum parameter for the powders prepared using the two synthetic methods. The findings reveal that the powder SGR-synthesized powder exhibits smaller and more uniform characteristics (0.68 times smaller crystal size) than its SSR counterpart. This discovery lays the groundwork for optimizing the powder manufacturing process of LLTO membranes, making them more suitable for various applications, including manufacturing high-performance membranes or mass production of membranes.
- [Korean]
- Development of Lithium Lanthanum Titanate (LLTO) Membrane Manufacturing Process for Selective Separation of Lithium Ion
- Young Il Kim, Sang Cheol Park, Kwang Ho Shin, InYeong Kim, Kee-Ahn Lee, Sung-Kyun Jung, Bin Lee
- J Powder Mater. 2023;30(1):22-28. Published online February 1, 2023
- DOI: https://doi.org/10.4150/KPMI.2023.30.1.22

- 3,055 View
- 50 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF The global demand for raw lithium materials is rapidly increasing, accompanied by the demand for lithiumion batteries for next-generation mobility. The batch-type method, which selectively separates and concentrates lithium from seawater rich in reserves, could be an alternative to mining, which is limited owing to low extraction rates. Therefore, research on selectively separating and concentrating lithium using an electrodialysis technique, which is reported to have a recovery rate 100 times faster than the conventional methods, is actively being conducted. In this study, a lithium ion selective membrane is prepared using lithium lanthanum titanate, an oxide-based solid electrolyte material, to extract lithium from seawater, and a large-area membrane manufacturing process is conducted to extract a large amount of lithium per unit time. Through the developed manufacturing process, a large-area membrane with a diameter of approximately 20 mm and relative density of 96% or more is manufactured. The lithium extraction behavior from seawater is predicted by measuring the ionic conductivity of the membrane through electrochemical analysis.
-
Citations
Citations to this article as recorded by- A Study on Particle and Crystal Size Analysis of Lithium Lanthanum Titanate Powder Depending on Synthesis Methods (Sol-Gel & Solid-State reaction)
Jeungjai Yun, Seung-Hwan Lee, So Hyun Baek, Yongbum Kwon, Yoseb Song, Bum Sung Kim, Bin Lee, Rhokyun Kwak, Da-Woon Jeong
journal of Korean Powder Metallurgy Institute.2023; 30(4): 324. CrossRef
- A Study on Particle and Crystal Size Analysis of Lithium Lanthanum Titanate Powder Depending on Synthesis Methods (Sol-Gel & Solid-State reaction)
TOP
 KPMI
KPMI


 First
First Prev
Prev


