Articles
- Page Path
- HOME > J Korean Powder Metall Inst > Volume 23(3); 2016 > Article
-
ARTICLE
- Fabrication and Mechanical Characteristics of Bulk Nickel/Carbon Nanotube Nanocomposites via the Electrical Explosion of Wire in Liquid and Spark Plasma Sintering Method
- Thuyet-Nguyen Minh, Hai-Nguyen Hong, Won Joo Kim, Ho Yoon Kim, Jin-Chun Kim*
-
Journal of Korean Powder Metallurgy Institute 2016;23(3):213-220.
DOI: https://doi.org/10.4150/KPMI.2016.23.3.213
Published online: May 31, 2016
School of Materials Science and Engineering, University of Ulsan, 93 Daehak-ro, Nam-gu, Ulsan 44610, Korea
- *Corresponding Author: Jin-Chun Kim, +82-52-259-2231, +82-52-259-1688, jckimpml@ulsan.ac.kr
• Received: May 13, 2016 • Revised: June 13, 2016 • Accepted: June 15, 2016
© The Korean Powder Metallurgy Institute. All rights reserved
- 1,619 Views
- 5 Download
- 2 Crossref
Figure & Data
References
Citations
Citations to this article as recorded by 

- Fabrication of nanocomposites by electric explosion of stainless steel capillaries filled with carbon nanotubes
Tao Jiang, Zhongyu Hou
Applied Surface Science.2020; 513: 145824. CrossRef - Effect of a nano-sized TiC particle addition on the flow-assisted corrosion resistance of SA 106B carbon steel
Jin-Ju Park, Eun-Kwang Park, Gyoung-Ja Lee, Chang-Kyu Rhee, Min-Ku Lee
Applied Surface Science.2017; 415: 143. CrossRef
Fabrication and Mechanical Characteristics of Bulk Nickel/Carbon Nanotube Nanocomposites via the Electrical Explosion of Wire in Liquid and Spark Plasma Sintering Method

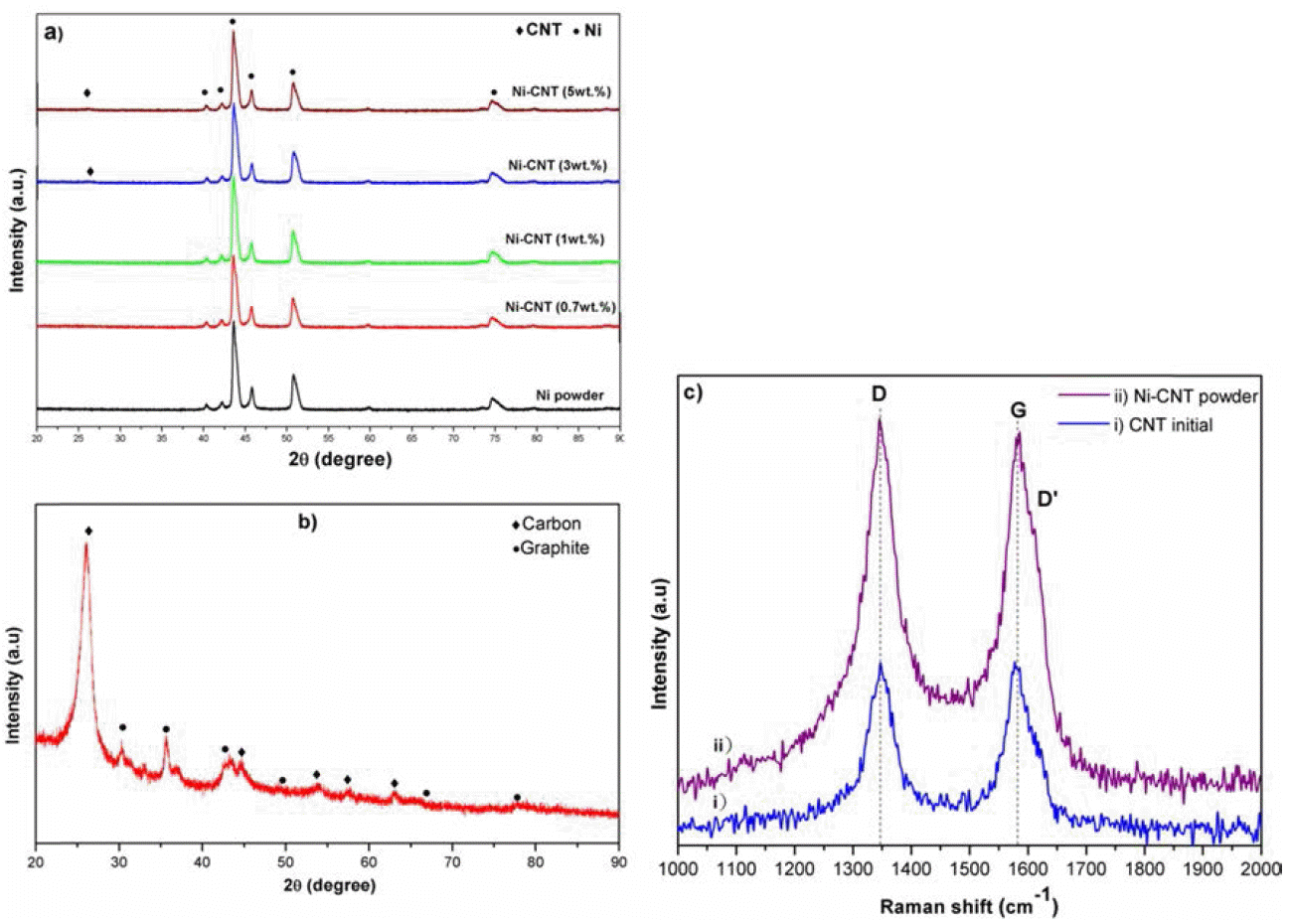



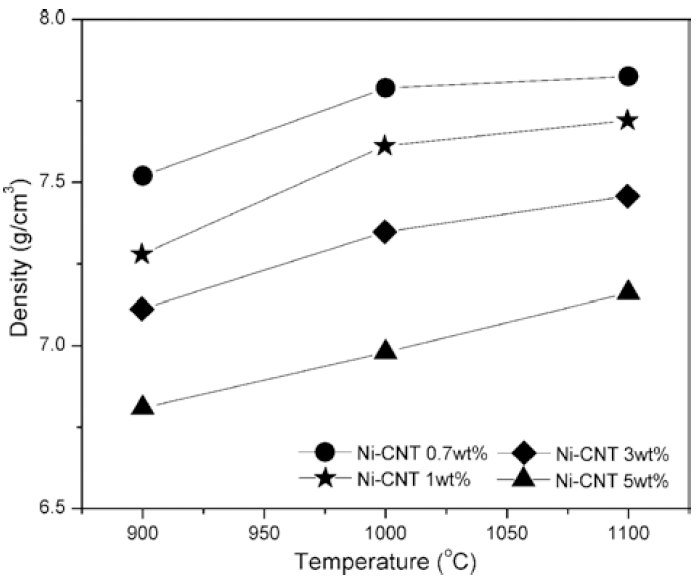
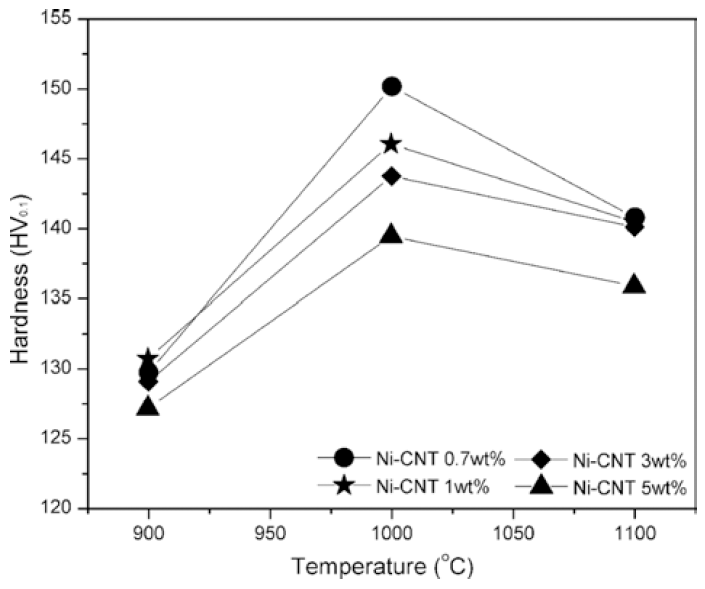
Fig. 1
FE-SEM images of (a) initial CNTs, (b) Ni powder, (c) Ni-0.7wt%CNT, (d) Ni-1wt%CNT, (e) Ni-3wt%CNT and (f) Ni-5 wt%CNT powders.
Fig. 2
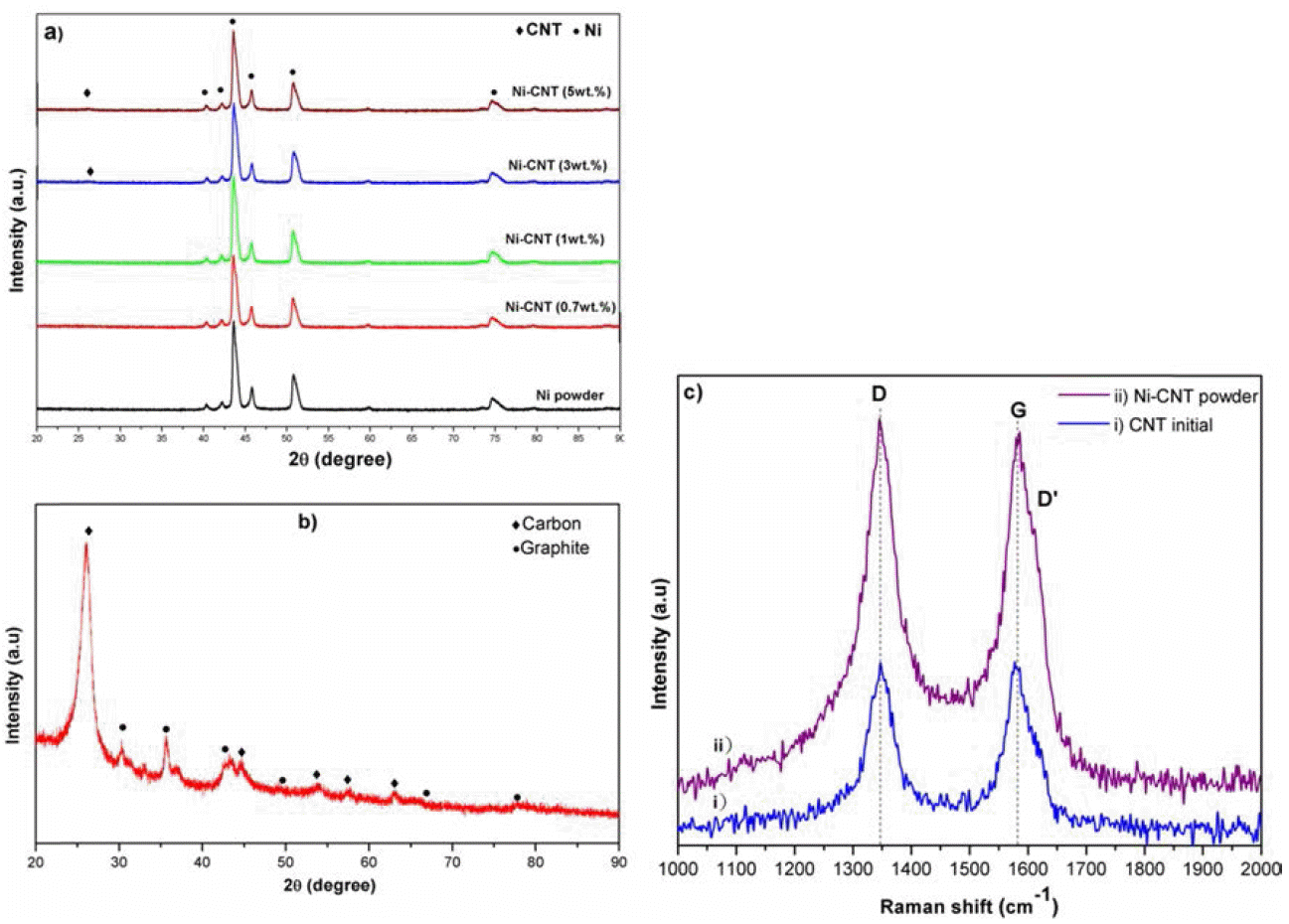
XRD patterns of the obtained (a) Ni powder, Ni-CNTs composite powders, (b) CNTs and (c) Raman spectra of CNTs and Ni-CNTs composite powder.
Fig. 3
Surface fracture of SPSed composite samples at 900°C: a) Ni-CNT 0.7wt.%; b) 1wt.%; c) 3wt.%; and d) 5wt.%.
Fig. 4
The optical microstructure of SPSed samples at 1000°C and 1100°C.
Fig. 5
EDS profiles of the dark region and white region in a Ni-CNTs composite after sintering.
Fig. 6
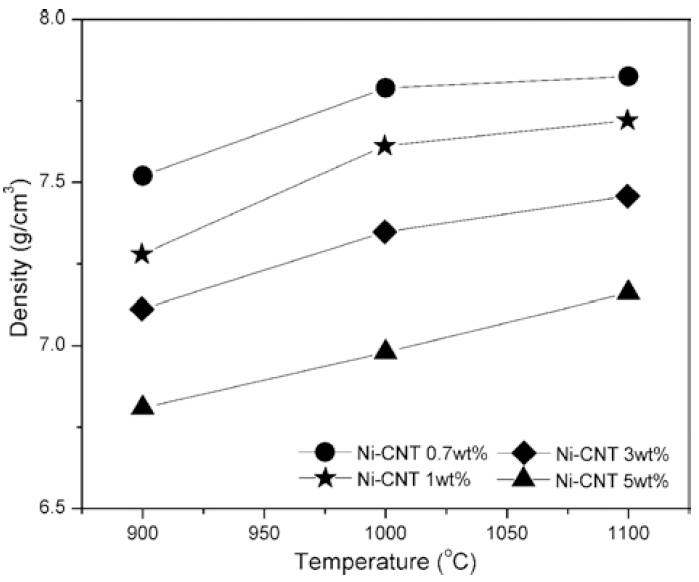
The density of SPSed samples at different temperature and with different content of CNTs.
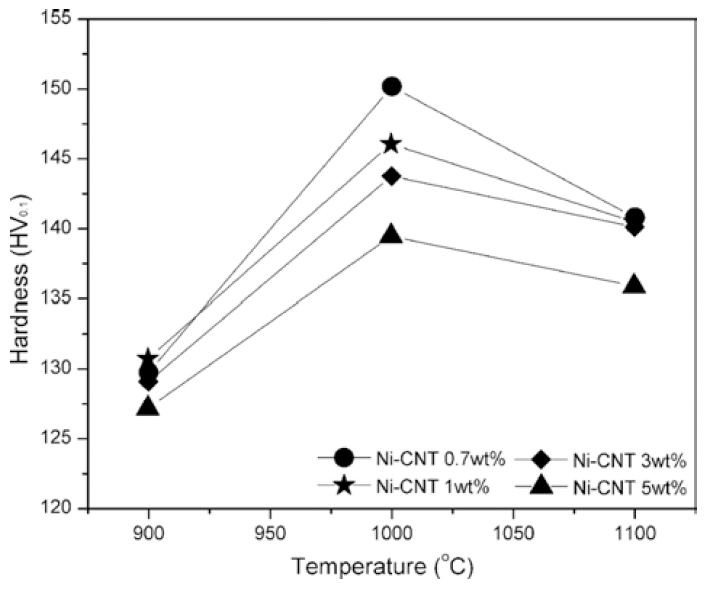
Fig. 7
The density of sintered body as a function of sintering temperature and the content of CNT.
Fig. 1
Fig. 2
Fig. 3
Fig. 4
Fig. 5
Fig. 6
Fig. 7
Fabrication and Mechanical Characteristics of Bulk Nickel/Carbon Nanotube Nanocomposites via the Electrical Explosion of Wire in Liquid and Spark Plasma Sintering Method
| Capacitance | 30 μF |
|---|---|
|
|
|
| Charging voltage | 30 KV |
| Ni wire diameter | 0.25 mm |
| Wire length of one explosion | 40 mm |
| Ambient liquid | Ethanol |
Table 1
Conditions of the electrical explosion of wire process
Table 1
TOP
 KPMI
KPMI





 Cite this Article
Cite this Article







