Articles
- Page Path
- HOME > J Powder Mater > Volume 30(1); 2023 > Article
-
Article
- Investigation on Microstructure and Flowability of Gas Atomized Heat-resistant KHR45A Alloy Powders for Additive Manufacturing
- Geonwoo Baeka, Mohsen Saboktakin Rizia, Yeeun Leea, SungJae Joa, Joo-Hyun Choib, Soon-Jik Honga,*
-
Journal of Korean Powder Metallurgy Institute 2023;30(1):13-21.
DOI: https://doi.org/10.4150/KPMI.2023.30.1.13
Published online: January 31, 2023
a Division of Advanced Materials Engineering, and Center for Advanced Materials and Parts of Powders (CAMP2), Kongju National University, 275, Budae-dong, Cheonan City 31080, Chungcheongnam-do, Republic of Korea
b Boogong Co. Ltd., 123-51, Injusandan-ro, Inju-myeon, Asan-si, Chungcheongnam-do, Republic of Korea
- * Corresponding Author: Soon-Jik Hong, TEL: +82-41-521-9387, FAX: +82-41-568-5776, E-mail: hongsj@kongju.ac.kr
- - G. Baek·M.S. Rizi: 학생, Y. Lee·S. J. Jo: 학생, J.-H. Choi: 연구원, S.-J. Hong: 교수
• Received: February 20, 2023 • Revised: February 23, 2023 • Accepted: February 23, 2023
© The Korean Powder Metallurgy Institute. All rights reserved.
- 3,074 Views
- 767 Download
- 2 Crossref
Abstract
- In additive manufacturing, the flowability of feedstock particles determines the quality of the parts that are affected by different parameters, including the chemistry and morphology of the powders and particle size distribution. In this study, the microstructures and flowabilities of gas-atomized heat-resistant alloys for additive manufacturing applications are investigated. A KHR45A alloy powder with a composition of Fe-30Cr-40Mn-1.8Nb (wt.%) is fabricated using gas atomization process. The microstructure and effect of powder chemistry and morphology on the flow behavior are investigated by scanning electron microscopy (SEM), X-ray diffraction (XRD), X-ray photoelectron spectroscopy (XPS), and revolution powder analysis. The results reveal the formation of spherical particles composed of single-phase FCC dendritic structures after gas atomization. SEM observations show variations in the microstructures of the powder particles with different size distributions. Elemental distribution maps, line scans, and high-resolution XPS results indicate the presence of a Si-rich oxide accompanied by Fe, Cr, and Nb metal oxides in the outer layer of the powders. The flowability behavior is found to be induced by the particle size distribution, which can be attributed to the interparticle interactions and friction of particles with different sizes.
- In industries such as aerospace, power generation, and chemical processing, there is a growing demand for materials that can withstand extreme temperatures and corrosive gas environments [1, 2]. High temperature corrosion can cause significant damage to materials, leading to equipment failures and costly downtime. Therefore, the development of materials that can operate in these harsh conditions is essential for improving the efficiency, safety, and reliability of these industries [3, 4]. To address these challenges, materials scientists and engineers are actively researching and developing new materials that can withstand high temperatures and corrosive environments [5].
- In this context, the first generation of austenitic heatresistant alloy composed of 25Cr-20Ni was introduced in the 1960s [6]. Heat-resistant KHR45A alloys are a type of high-temperature alloy that are designed to maintain their strength and toughness in extreme heat conditions. Heat-resistant KHR45A alloys are an important class of materials that play a critical role in the design and operation of high-temperature equipment in a range of industries. They are commonly used in applications such as gas turbines, steam turbines, and other high-temperature equipment in the power generation, petrochemical, and aerospace industries. KHR45A alloys are made with a specific chemical composition that includes high levels of Cr and Ni, which provide excellent resistance to oxidation and corrosion at high temperatures [5-7]. Additionally, they are strengthened by the presence of other elements such as Mo, W, V and Nb which contribute to their high temperature stability and mechanical strength. These alloys are known for their ability to withstand temperatures up to 1100°C without losing their strength and durability [2]. They also have good creep resistance, meaning they can maintain their shape and strength under prolonged stress at high temperatures. For instance, KHR45A alloy tubes are commonly used in ethylene cracking furnaces due to their excellent high-temperature strength, thermal conductivity, and resistance to oxidation and carburization. However, the cracking process can generate coke, which can deposit onto the inner surface of the tubes, causing them to degrade and fail over time. The presence of a protective oxide layer, particularly Cr2O3, can inhibit the adsorption of coke onto the inner surface of the tube and, therefore, keep carbon from easily intruding into the metal. When KHR45A alloy tubes are exposed to high temperatures in an ethylene cracking furnace, an oxide layer naturally forms on their surface. This oxide layer acts as a barrier between the alloy and the environment, reducing the rate of oxidation and carburization of the metal. In the presence of chromium, which is a common alloying element in KHR45A alloy, the oxide layers that forms are primarily composed of Cr2O3. This oxide layer is particularly effective at inhibiting coke adsorption onto the inner surface of the tubes, as it has a low surface energy and is hydrophobic, which makes it difficult for coke to adhere to the surface [5, 8]. The presence of a protective oxide layer on KHR45A alloy tubes can significantly increase their service life and reduce the need for frequent maintenance and replacement. In addition, it can improve the overall efficiency and safety of the ethylene cracking furnace by reducing the risk of tube failure and minimizing the release of harmful gases and pollutants into the environment. However, it is important to note that the formation and maintenance of the oxide layer require careful control of the operating conditions, including temperature, gas composition, and the presence of impurities such as sulfur and phosphorus. Furthermore, the oxide layer can be damaged by mechanical or thermal stresses, which can lead to accelerated tube degradation and failure. Therefore, regular inspection and maintenance are essential to ensure the protective oxide layer remains intact and effective over time. Traditional methods for producing heat-resistant alloys typically involve casting, forging, or powder metallurgy. These methods involve melting or forming the alloy into a desired shape, followed by controlled cooling and heat treatment to achieve the desired mechanical properties [9]. The specific method used depends on the type of alloy being produced, the desired properties, and the intended application. In contrast, additive manufacturing (AM), also known as 3D printing, is a relatively new method for producing heat-resistant alloys. It involves the layer-by-layer deposition of metal powder or wire using a high-energy heat source, such as a laser or electron beam. This allows for the production of complex geometries and internal features that are difficult or impossible to achieve using traditional methods [10, 11]. One advantage of AM for heat-resistant alloys production is the ability to produce parts with tailored microstructures and properties, which can improve their hightemperature performance. For example, AM can produce parts with fine-grained structures, which can enhance their strength and toughness. In addition, AM can be used to produce parts with graded or functionally graded structures, which can optimize their performance in specific areas or under different loading conditions. Another advantage of AM is its ability to produce small batches of parts or prototypes quickly and cost-effectively, without the need for expensive tooling or machining [12, 13]. This can be particularly beneficial for the development and testing of new heat-resistant alloys or designs, as it allows for rapid iteration and optimization. However, there are also some challenges associated with using AM for heat-resistant alloys production. One key challenge is the need to optimize the process parameters and control the microstructure and properties of the parts, which can be affected by factors such as powder characteristics, laser power, and scanning strategy. This paper aims at studying the properties and microstructure of gas atomized KHR45A alloys powders particularly focusing on powder flowability for additive manufacturing applications.
1. Introduction
- This work investigates the processability of Cr-Ni heatresistant alloy by gas atomization and its potential for AM process applications. For this purpose, KHR45A alloy ingot were prepared by casting with the chemical composition reported in Table 1. The process started by melting the high purity elements (purity higher than >99.9%) in an induction furnace under argon atmosphere at above 1500°C. Subsequently, the gas atomization (CAMP2) was conducted in a gas atomizer chamber and the alloy melt flowed through a boron-nitride delivery nozzle with hole size of 8 mm using nitrogen gas at a pressure of 1 MPa and were collected at the bottom of the chamber. A laser particle size analyzer was used to measure the particle size distribution of the atomized powders (Malvern Panalytical Mastersizer 3000E). To identify the phases structure of the gas atomized KHR45A alloy powders, X-ray diffraction (XRD) analysis was performed using a MiniFlex600, Rigaku, Japan device with Cu-Kα radiation (λ = 1.54056 Å, 2θ range from 20° to 100°, step size of 0.02° and dwell time of 1s per step at 40 kV and 200 mA). The obtained XRD patterns of the powders were processed with X’pert High- Score software. The surface morphology of gas atomized KHR45A alloy powders were examined using scanning electron microscopy (SEM, TESCAN, MIRA LMH) equipped with energy dispersive spectrometer (EDS). The surface chemical composition and electronic structure were characterized using X-ray photoelectron spectroscopy (XPS; Thermo VG Scientific, K-Alpha) with the C 1s peak (C–C bond) at 285.0 eV serving as the calibration. The flowability of the gas atomized powders was quantitatively measured using a revolution powder analyzer (CAMP2: Mercury Scientific Inc., Newtown, CT, USA).
2. Experimental
- Since the size and morphology of powders affects the performance of feedstock materials in additive manufacturing processes, the morphology and particle size distribution of KHR45A alloy powders investigated in this study were studied by SEM and a laser particle size analyzer. The SEM images of the powder particle morphologies prepared by gas atomization are shown in Fig. 1(ac). As can be observed from Fig. 1 (a), the gas-atomized KHR45A alloy powders are composed of fine and large particles, which particles typically are spherical in shape and have a smooth surface. As a result of filling in voids between the larger particles, the packing density in a material could be increased by presence of high-volume fraction of fine particles [14]. In general, shape factors including the circularity, solidity, convexity, etc., can be used to evaluate particle shape [15]. Finer particles, as shown in Fig. 1 (b), exhibit higher circularity compared to those of larger particles and only a few satellite particles are observed in fine particles. Furthermore, top view microstructure observation revealed that KHR45A powders had very fine internal structure composed of dendrite- like structures and multiple dendrite arms are visible. Pore structures corresponding to the shrinkage behavior of high viscosity melt during the solidification were not observed in this magnification. However, there are a greater number of satellites connected to the larger particles (Fig. 1 (c)), which results in a lower circularity. By considering each larger particle and its satellites as one particle, the shape of the particle would be different from the shape of a circle. The formation of satellites on the surfaces of larger atomized powders can be attributed to the greater surface area and roughness of the larger particles [16]. During gas atomization, molten droplets solidify into irregularly shaped particles with varying sizes and surface roughness. The roughness of the particle surface can promote the adhesion of smaller, satellitelike particles that are produced during the gas atomization process. It is worth noting that the factors influencing satellite formation are complex, and the size of the gas atomized powders is just one of many factors that can affect the formation of satellites. Other factors, such as processing conditions, material properties, and the characteristics of the gas atomization system, can also influence the formation of satellites. It is generally believed that high circularity results in beneficial flow properties. On the other hand, satellite particles can act as roughness or asperities on the surface of the powder particles, leading to increased friction and reduced powder flowability. In addition, the presence of satellite particles on the surface of powder particles can also reduce the packing density of the powder or satellite particles can interfere with the sintering process, leading to reduced density, inferior mechanical properties, reduced strength and increased porosity in the final product [17, 18]. The particle size distribution is presented in Fig. 1 (d). Under the current gas atomization condition, the mean gasatomized particle size of d10, d50 and d90 were measured to be 61 μm, 101 μm and 165 μm respectively.
- Fig. 2 shows the XRD patterns of the KHR45A gasatomized powders. The diffraction peaks of the KHR45A gas-atomized powders belong to a single-phase FCC structure. The diffraction peaks are approximately at 2θ values of 35.4°, 44.41°, 50.81°, 74.66°, 90.64°, 95.92°, which correspond to (110), (111), (200), (220), (311) and (222) planes of the FCC structures, respectively. No diffraction peaks related to other phases such as secondary phases or MC type carbides were detected, suggesting that the solid solution FCC phase was formed after gas atomization process. In order to calculate the lattice parameter Rietveld refinement analysis was performed and the lattice constant of gas-atomized KHR45A alloy which calculated based on the average peak angles of (111) planes was measured to be 3.56 Å This value is consistent with those reported for Fe-Cr-Ni alloys [19].
- In order to understand the morphology and structure of powders after gas atomization, cross-section microstructures of the powders after polishing and etching in a solution of 1 ml HNO3 and 3 ml HCl were investigated by SEM and EDS map. Fig. 3 (a-c) shows the cross-sectional morphology and structure of gas atomized KHR45A alloy powders with different particle size of 50 μm, 80 μm and 120 μm respectively. As can be seen in Fig. 3 (a-c) the microstructure of the particles with different size represents the typical dendritic microstructure of the ascast alloys, but noticeable difference is microstructural features of the three particles. Larger particles, however, show an increasing amount of segregation in the interdendritic regions (as shown in Fig. 3 (c1)). It has been reported that cooling rate and particle size are important factors in the formation of dendritic microstructure. It is to be expected that slower cooling rate could be applied on larger particles, which would result in segregation of elements adjacent to the interdendritic regions [20]. The cooling rate and dendritic structure evolution of gas atomized powders can be affected by the particle size as follows: (i) Surface area-to-volume ratio: as the particle size decreases, the surface area-to-volume ratio of the powder increases. This means that smaller particles have a larger surface area per unit volume, which can lead to more rapid cooling rates due to increased heat transfer to the surrounding gas. (ii) Heat dissipation: smaller particles can also cool faster due to their ability to dissipate heat more efficiently. This can result in finer dendritic structures in the solidified powders. (iii) Increased undercooling: smaller particles can experience greater undercooling, which is the difference between the equilibrium solidification temperature and the actual temperature at which solidification occurs. This can lead to the formation of metastable phases or the suppression of certain phases, which can affect the dendritic structure of the powder. Overall, decreasing the particle size of gas-atomized powders can result in faster cooling rates and finer dendritic structures, which can have implications for the mechanical and physical properties of the final product.
- The SEM image and corresponding elemental distribution map accompanied by line scan of dendritic structure was presented in Fig. 4 (a1-a3). The corresponding EDS maps in Fig. 4 (a3) proved homogenous distribution of Fe, Ni, Cr, Mn, Nb and Si alloying elements at inner layer of powder while, the high concentration of Si and O was observed at outer surface layer of gas atomized KHR45A alloy powder, which suggesting formation of SiO2 oxide at the outer surface layer. The average thickness of SiO2 oxide layer was measured to be 0.6 μm. It has been demonstrated that during the gas atomization of alloys containing Si, formation of SiO2 can be effectively promoted by high temperature, partial pressure of oxygen in the gas stream and higher affinity of Si with oxygen. The composition profiles of EDS line scan results along the A–B line in Fig. 4 (a1) also suggest the segregation of Cr at interdendritic regions.
- Chemical state and elemental composition of gas atomized KHR45A alloy powder were analyzed by XPS. The XPS survey spectra analysis in Fig. 5 (a) confirmed the coexistence of Nb, Cr, Mn, Fe, Ni, Si and O elements. The high-resolution core level spectra of Nb3d, Cr2p, Mn2p, Fe2p, Ni2p, Si2p and O1s after fitting are shown in Fig. 5 (b-h). Spectra of the KHR45A powder fabricated by gas atomization show the contribution of all components. Two intense symmetrical of Cr2p1/2 and Cr2p3/2, Fe2p1/2 and Fe2p3/2, Ni2p1/2 and Ni2p3/2, Mn2p1/2 and Mn2p3/2, peaks were observed in the core-level spectra of Cr2p, Fe2p, Ni2p and Mn2p respectively (Fig. 5(cf)), while two intense symmetrical of Nb3d3/2 and Nb3d5/2 were found in the core-level spectra of Nb3d (Fig. 5 (b)). Presence of asymmetrical peaks which observed in the core-level spectra of Cr, Fe, Ni, Mn and Nb revealed that these elements in gas atomized KHR45A alloy powder were in solid solution state. Furthermore, the XPS results confirmed the presence of metal oxides including Nb2O5, FeCr2O4, NiCr2O4, MnFeCr2O4 and SiO2 on the surface of gas atomized powder. It has been reported that presence of high chromium content can reduce the amount of silicon required for oxide formation. On the other hand, high affinity of Si with oxygen compared to other elements such as Cr which depends on the valence electrons is known to enhance the formation of SiO2 during the gas atomization process [21-23].
- Fig. 6 presents the flowability behavior of gas-atomized KHR-45A powder with three different size distributions of 50-100 μm, 100-150 μm, and 50-150 μm. Fig. 6 (a) compares the avalanche energy of these three size distributions, which is the minimum energy required to initiate a localized avalanche flow of the powder. Results indicate that the powders with the size of 50-100 μm have the highest avalanche energy, while the powder in the range of 100-150 μm has the lowest, and the powders distributed in the range of 50-150 μm have an intermediate value. Particle size is a primary factor that affects powder flowability. Smaller particles have a higher surface area to volume ratio and tend to have more interparticle interactions, leading to increased friction and difficulty in flowing smoothly. Larger particles, on the other hand, are less prone to these interactions and flow more easily. Thus, the powders in the range of 50-100 μm with a higher proportion of smaller particles has a higher avalanche energy due to more interparticle interactions and an increased tendency to form localized avalanche flows. The powders in the range of 100-150 μm with a lower proportion of smaller particles has a lower avalanche energy. The particles within the range of 50- 150 μm, with a balanced ratio of larger and smaller particles, has an intermediate avalanche energy due to a balance between surface area and volume effects on particle interactions and flow behavior [24].
- In Fig. 6 (b), the avalanche angle and rest angle of the powders are presented. The particles within the range of 50-100 μm represented the lowest avalanche and rest angles due to smaller particles forming a more stable pile and resisting flow until a steeper angle of inclination is reached. The particles with the size of 100-150 μm have a higher avalanche and rest angle, likely due to the presence of a larger proportion of larger particles that are more prone to interparticle interactions and less stable packing. Furthermore, the powder in the range of 50-150 μm represent a more balanced distribution of particle sizes, resulting in a more stable packing arrangement and similar avalanche and rest angles compared to those of particles in the range of 100-150 μm.
- Fig. 6(c) displays the avalanche energy vs. avalanche time graph of the powders, which shows the relationship between the energy required to initiate an avalanche flow and the time it takes for the flow to propagate. The graph has two regions: an initial low-energy regime where the powder is stable and a high-energy regime where the powder undergoes an avalanche flow. Powders with a steep slope (powders within the range of 50-150 μm) have a rapid increase in energy as the flow propagates and may be more likely to cause clogs or blockages. Powders with a shallower slope (i.e., particles in the range of 50-100 μm) indicate a slower increase in energy and smoother flow.
- Fig. 6 (d) shows the avalanche time graph of the powders. The powders within the range of 50-100 μm has the maximum time due to the high degree of interparticle interactions and friction caused by a larger proportion of smaller particles. However, the powder in the range of 50-150 μm has a more balanced distribution of particle sizes, resulting in a smoother and faster flow behavior. On the other hand, the 100-150 μm sized powder has a flow behavior that is intermediate between the other two powders.
- Fig. 6 (e) shows the cumulative frequency vs. avalanche angle graph of the powders. The graph has an Sshaped curve, in which the particles within the range of 50-100 μm display more gradual increase (from 27.6° to 29.5° degree) in cumulative frequency as the angle of inclination increases, indicating smoother flow behavior. The other particles in the size range of 100-150 μm and 50-150 μm represent steeper initial slopes (29° to 31.5°), suggesting they are more prone to clogging or blockages and may have a more difficult time flowing smoothly.
3. Results and Discussion
Fig. 1

(a) The microstructure and morphology of the gas atomized KHR45A alloy powders, (b and c) Top view of particles with the diameter of (b) 75 μm and (c) 152 μm, (d) particle size distribution.

Fig. 3

SEM cross section observation of gas atomized KHR45A alloy powders in different particle size range of (a1) 50 μm, (b1) 80 μm, (c1) 120 μm, (a2, b2, c2) the enlarged SEM image of the rectangular regions in (a1, b1, c1) respectively.

Fig. 4

SEM micrographs illustrating the (a1, a2) dendritic microstructure of the gas atomized KHR45A alloy powders, (a3) acquired elemental distribution maps and line scan exhibiting concentrations of constituent elements in the KHR45A powder which display formation of Si rich oxide at the surface of powder.

Fig. 5

High-resolution XPS results for Nb, Cr, Mn, Fe, Ni, Si and O core-levels of gas atomized KHR45A alloy powder.

Fig. 6

Flowability behavior of gas atomized KHR45A alloy powder, (a) avalanche energy, (b) avalanche angle and rest of angle, (c) avalanche energy vs. avalanche time, (d) avalanche time vs. particle size, (e) cumulative frequency vs. avalanche angle.

- This study investigated the microstructure and flowability behavior of gas-atomized KHR45A alloy powders for additive manufacturing applications. It was found that the powders consist of an FCC phase and displayed a dendritic microstructure. The SEM–EDS results indicated the homogenous distribution of constituent alloying elements at inner layer of powder while, the high concentration of Si and O was observed at outer surface layer of gas atomized KHR45A alloy powder. The XPS results confirmed the presence of metal oxides including Nb2O5, FeCr2O4, NiCr2O4, MnFeCr2O4 and SiO2 on the surface of gas atomized powder. The flowability of the powders was found to be affected by particle size distribution, the highest avalanche energy of 1.5 mJ/kg reported for particles in the range of 50-100 μm while the particles in the range of 100-150 μm exhibited the lowest values. The cumulative frequency vs. avalanche angle graph showed an S-shaped curve for all three powder size distributions, with the powders in the range of 50-100 μm resulted in the smoothest flow behavior. It is expected that our work provides insight into the factors that influence the flowability behavior of gasatomized KHR45A alloy during the additive manufacturing process.
4. Conclusions
-
Acknowledgements
- This work was supported by the Korea Institute for Advancement of Technology (KIAT), and Ministry of Trade, Industry and Energy (P0018009). This work was supported by the Technology development Program (S3211124) funded by the Ministry of SMEs and Startups (MSS, Korea).
Acknowledgments
- 1. M. Ebrahimian, M. H. Enayati, F. Karimzadeh, Y. Min and D.-E. Kim: Mater Charact., 171 (2021) 110816..Article
- 2. A. Delić, M. Oruč, M. Rimac, A. Kump and P. Anderson: Simulation Centrifugal Casting of the Heat Resistant Austenitic Steel HK 30 Modified by Niobium, Springer Cham, 76 (2020) 146..Article
- 3. V. Tari, A. Najafizadeh, M. H. Aghaei and M. A. Mazloumi: Journal of Failure Analysis and Prevention., 9 (2009) 316..Article
- 4. A. El-Batahgy and B. Zaghloul: Mater Charact., 54 (2005) 239..Article
- 5. A. Amanov, J.-H. Choi and Y.-S. Pyun: Materials, 14 (2021) 3658..ArticlePubMedPMC
- 6. A. A. Kaya: Mater Charact., 49 (2002) 23. .Article
- 7. L. M. Shen, J. M. Gong and H. S. Liu: Materials at High Temperatures., 31 (2014) 148..Article
- 8. H.-J. Kim, J. Park, Y.-S. Ji, B.M. Jeong, J. Nam, M.-G. Jo, J. Lee, D.-I. Kim, J.-Y. Suh and J.-H. Shim: Mater Charact., 176 (2021) 111110..Article
- 9. Y. Lee, C. Nagarjuna, J.-W. Song, K.Y. Jeong, G. Song, J. Lee, J.-H. Lee and S.-J. Hong: Powder Metallurgy., 64 (2021) 219..Article
- 10. G. Shin, M. Ebrahimian, N. K. Adomako, H. Choi, D. J. Lee, J.-H. Yoon, D. W. Kim, J.-Y. Kang, M. Y. Na, H. J. Chang and J. H. Kim: Mater Des., 227 (2023) 111681..Article
- 11. W. Choo, M. Ebrahimian, K. Choi and J. H. Kim: Metals and Materials International., (2022)..
- 12. M. M. Francois, A. Sun, W. E. King and N. J. Henson: Curr Opin Solid State Mater Sci., 21 (2017) 198..Article
- 13. M. Molitch-Hou: Addit Manuf., (2018) 1..Article
- 14. T. Fedina, J. Sundqvist, J. Powell and A. F. H. Kaplan: Addit Manuf., 36 (2020) 101675..Article
- 15. J. Boes, A. Röttger and W. Theisen: Addit Manuf., 34 (2020) 101379..Article
- 16. D. Beckers, N. Ellendt and U. Fritsching: Advanced Powder Technology., 31 (2020) 300..Article
- 17. S. Özbilen: Powder Metallurgy., 42 (1999) 70..Article
- 18. L.-C. Zhang, W.-Y. Xu and Z. Li: Powder Technol., 418 (2023) 118162..Article
- 19. C. Wu, B.-J. Lee and X. Su: Calphad., 57 (2017) 98..Article
- 20. F. Pengjun, X. Yi, L. Xinggang and C. Ya: Rare Metal Materials and Engineering., 47 (2018) 423..Article
- 21. H. Buscail, C. Issartel and F. Riffard: Corros Sci., 65 (2012) 535..Article
- 22. A. Barroux, T. Duguet and N. Ducommun: Surfaces and Interfaces., 22 (2021) 100874..Article
- 23. R. Oro, M. Campos and E. Hryha: Mater Charact., 86 (2013) 80..Article
- 24. P. Kiani, U. Scipioni Bertoli and A. D. Dupuy: Adv Eng Mater., 22 (2020) 2000022..Article
Figure & Data
References
Citations
Citations to this article as recorded by 

- Engineering heterogeneous microstructure for enhancing mechanical properties of multicomponent alloys via powder metallurgy route
Min Woo Shin, Sung-Jae Jo, Sourabh Kumar Soni, Ji-Woon Lee, Jongun Moon, Hyoung Seop Kim, Soon-Jik Hong
Materials Science and Engineering: A.2025; 941: 148599. CrossRef - Al-based amorphous coatings by warm spraying: Numerical simulation and experimental validation
Deming Wang, Nianchu Wu, Peng Cao
Journal of Alloys and Compounds.2024; 1008: 176674. CrossRef
Investigation on Microstructure and Flowability of Gas Atomized Heat-resistant KHR45A Alloy Powders for Additive Manufacturing

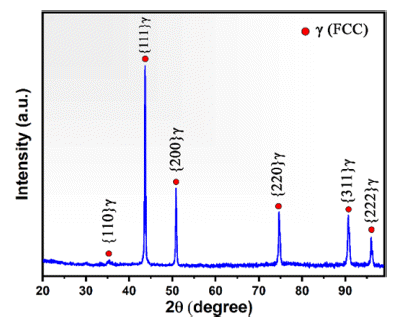

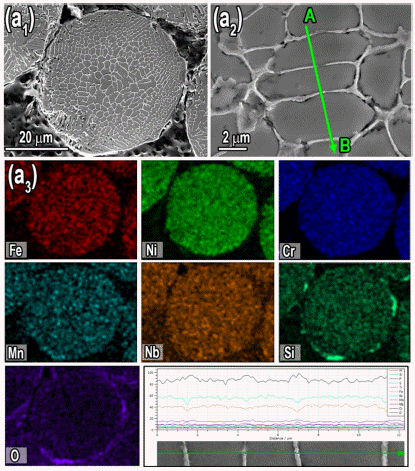
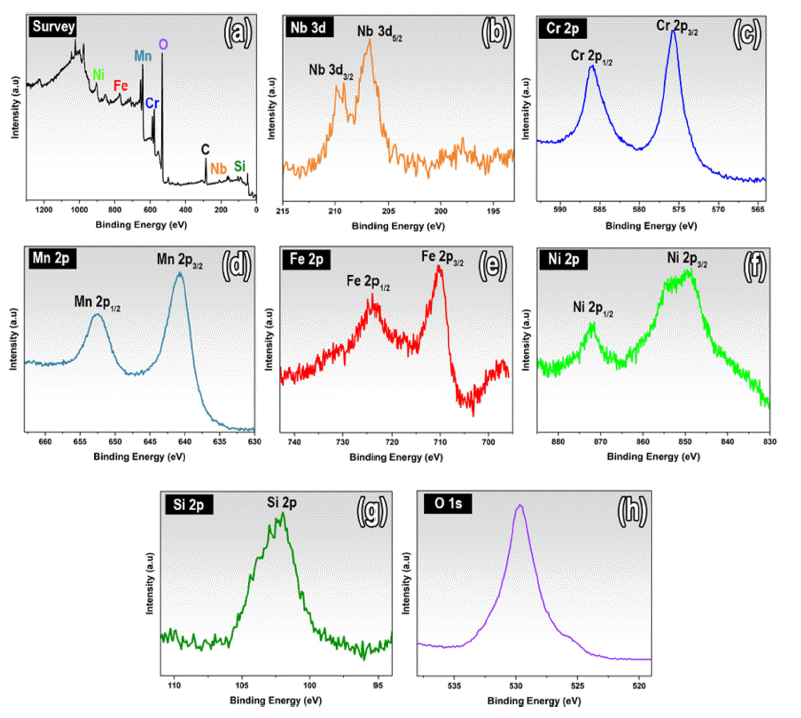
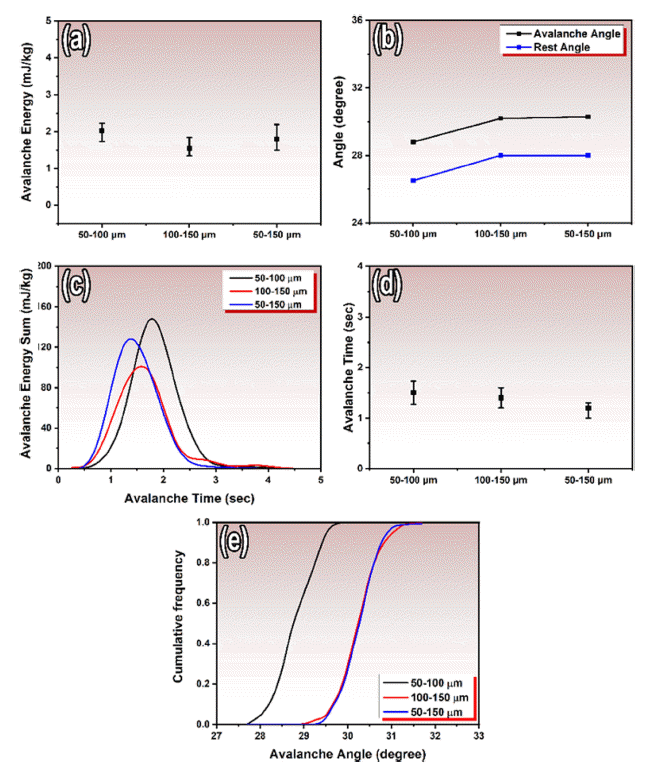
Fig. 1
(a) The microstructure and morphology of the gas atomized KHR45A alloy powders, (b and c) Top view of particles with the diameter of (b) 75 μm and (c) 152 μm, (d) particle size distribution.
Fig. 2
XRD patterns of the gas atomized KHR45A alloy powders.
Fig. 3
SEM cross section observation of gas atomized KHR45A alloy powders in different particle size range of (a1) 50 μm, (b1) 80 μm, (c1) 120 μm, (a2, b2, c2) the enlarged SEM image of the rectangular regions in (a1, b1, c1) respectively.
Fig. 4
SEM micrographs illustrating the (a1, a2) dendritic microstructure of the gas atomized KHR45A alloy powders, (a3) acquired elemental distribution maps and line scan exhibiting concentrations of constituent elements in the KHR45A powder which display formation of Si rich oxide at the surface of powder.
Fig. 5
High-resolution XPS results for Nb, Cr, Mn, Fe, Ni, Si and O core-levels of gas atomized KHR45A alloy powder.
Fig. 6
Flowability behavior of gas atomized KHR45A alloy powder, (a) avalanche energy, (b) avalanche angle and rest of angle, (c) avalanche energy vs. avalanche time, (d) avalanche time vs. particle size, (e) cumulative frequency vs. avalanche angle.
Fig. 1
Fig. 2
Fig. 3
Fig. 4
Fig. 5
Fig. 6
Investigation on Microstructure and Flowability of Gas Atomized Heat-resistant KHR45A Alloy Powders for Additive Manufacturing
Table 1
Chemical compositions of gas atomized KHR45A alloy powders (wt%)
Table 1
TOP
 KPMI
KPMI



 Cite this Article
Cite this Article






