Search
- Page Path
- HOME > Search
- [English]
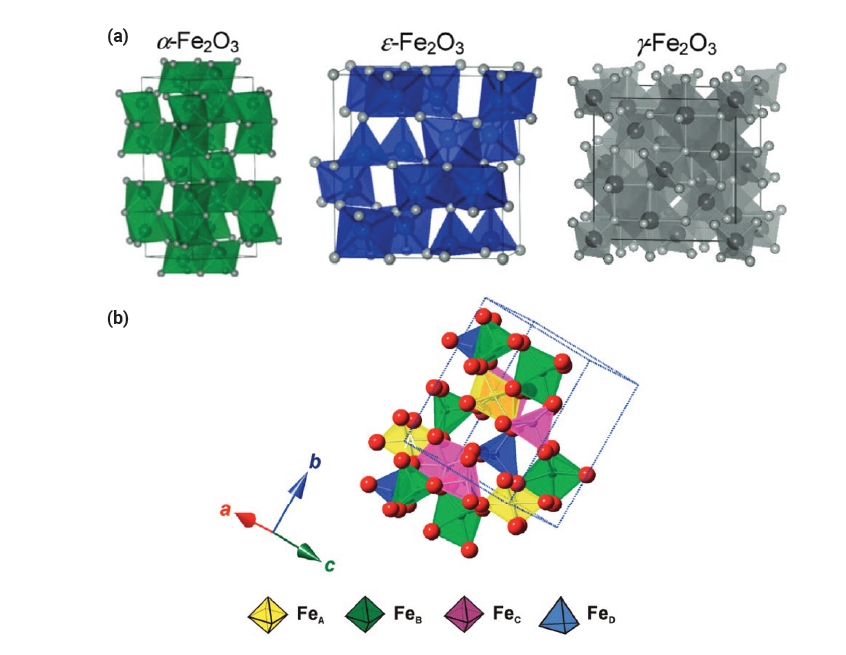
- Epsilon Iron Oxide (ε-Fe2O3) as an Electromagnetic Functional Material: Properties, Synthesis, and Applications
- Ji Hyeong Jeong, Hwan Hee Kim, Jung-Goo Lee, Youn-Kyoung Baek
- J Powder Mater. 2024;31(6):465-479. Published online December 31, 2024
- DOI: https://doi.org/10.4150/jpm.2024.00290
- 4,793 View
- 125 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF - Iron oxide (ε-Fe₂O₃) is emerging as a promising electromagnetic material due to its unique magnetic and electronic properties. This review focuses on the intrinsic properties of ε-Fe₂O₃, particularly its high coercivity, comparable to that of rare-earth magnets, which is attributed to its significant magnetic anisotropy. These properties render it highly suitable for applications in millimeter wave absorption and high-density magnetic storage media. Furthermore, its semiconducting behavior offers potential applications in photocatalytic hydrogen production. The review also explores various synthesis methods for fabricating ε-Fe₂O₃ as nanoparticles or thin films, emphasizing the optimization of purity and stability. By exploring and harnessing the properties of ε-Fe₂O₃, this study aims to contribute to the advancement of next-generation electromagnetic materials with potential applications in 6G wireless telecommunications, spintronics, high-density data storage, and energy technologies.
-
Citations
Citations to this article as recorded by- A Comprehensive Review of GPR Data Analysis for Bridge Deck Evaluation: From Conventional Methods to Emerging Artificial Intelligence Approaches
Babak Enami Alamdari, Yu Tang, Danilo Erricolo, Lesley H. Sneed
Journal of Nondestructive Evaluation.2026;[Epub] CrossRef - Chemical Pressure Induced Strain Control of Magnetic Anisotropy in the Simple Perovskite ϵ-Fe2O3
Subir Roy, Gurleen K. Uppal, Alberto Acosta, Rachel Nickel, Charles A. Roberts, Johan van Lierop
Nano Letters.2026; 26(1): 34. CrossRef - Superparamagnetism of Baked Clays Containing Polymorphs of Iron Oxides: Experimental Study and Theoretical Modeling
Petr Kharitonskii, Andrei Krasilin, Nadezhda Belskaya, Svetlana Yanson, Nikita Bobrov, Andrey Ralin, Kamil Gareev, Nikita Zolotov, Dmitry Zaytsev, Elena Sergienko
Magnetochemistry.2025; 11(12): 103. CrossRef
- A Comprehensive Review of GPR Data Analysis for Bridge Deck Evaluation: From Conventional Methods to Emerging Artificial Intelligence Approaches
- [Korean]
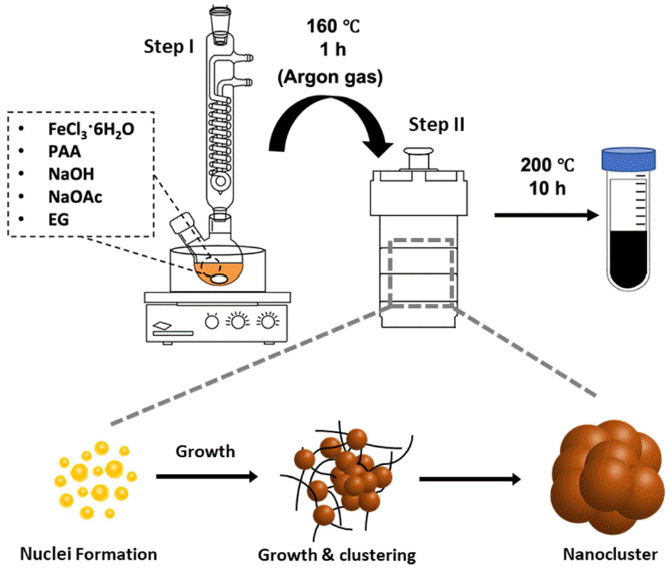
- Size Control of Iron Oxide (Fe3O4) Nanoclusters according to Reaction Factors and Consequent Change in Their Magnetic Attraction
- Sanghoon Lee, Arim Byun, Jin-sil Choi
- J Powder Mater. 2023;30(4):297-304. Published online August 1, 2023
- DOI: https://doi.org/10.4150/KPMI.2023.30.4.297
- 1,319 View
- 16 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF Iron oxide (Fe2O3) nanoclusters exhibit significant potential in the biomedical and pharmaceutical fields due to their strong magnetic properties, stability in solutions, and compatibility with living systems. They excel in magnetic separation processes, displaying high responsiveness to external magnetic fields. In contrast to conventional Fe2O3 nanoparticles that can aggregate in aqueous solutions due to their ferrimagnetic properties, these nanoclusters, composed of multiple nanoparticles, maintain their magnetic traits even when scaled to hundreds of nanometers. In this study, we develop a simple method using solvothermal synthesis to precisely control the size of nanoclusters. By adjusting precursor materials and reducing agents, we successfully control the particle sizes within the range of 90 to 420 nm. Our study not only enhances the understanding of nanocluster creation but also offers ways to improve their properties for applications such as magnetic separation. This is supported by our experimental results highlighting their size-dependent magnetic response in water. This study has the potential to advance both the knowledge and practical utilization of Fe2O3 nanoclusters in various applications.
-
Citations
Citations to this article as recorded by- Enzymatic properties of iron oxide nanoclusters and their application as a colorimetric glucose detection probe
Dahyun Bae, Minhee Kim, Jin-sil Choi
RSC Advances.2025; 15(6): 4573. CrossRef
- Enzymatic properties of iron oxide nanoclusters and their application as a colorimetric glucose detection probe
- [Korean]
- Controlling the Heat Generation Capability of Iron Oxide-Base Nanoparticles
- Jin-sil Choi
- J Korean Powder Metall Inst. 2021;28(6):518-526. Published online December 1, 2021
- DOI: https://doi.org/10.4150/KPMI.2021.28.6.518

- 838 View
- 7 Download
-
 Abstract
Abstract
 PDF
PDF This review summarizes the recent progress in iron-oxide-based heat generators. Cancer treatment using magnetic nanoparticles as a heat generator, termed magnetic fluid hyperthermia, is a promising noninvasive approach that has gained significant interest. Most previous studies on improving the hyperthermia effect have focused on the construction of dopant-containing iron oxides. However, their applications in a clinical application can be limited due to extra dopants, and pure iron oxide is the only inorganic material approved by the Food and Drug Administration (FDA). Several factors that influence the heat generation capability of iron-oxide-based nanoparticles are summarized by reviewing recent studies on hyperthermia agents. Thus, our paper will provide the guideline for developing pure iron oxide-based heat generators with high heat dissipation capabilities.
- [Korean]
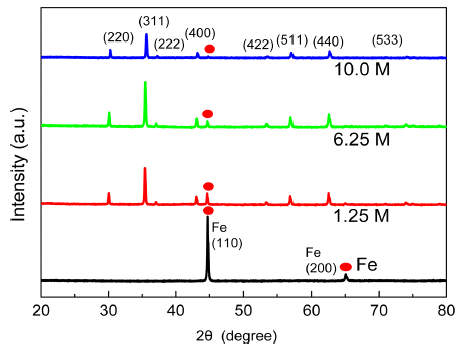
- Magnetic Properties of Micron Sized Fe3O4 Crystals Synthesized by Hydrothermal Methods
- Ki-Bum Lee, Chunghee Nam
- J Korean Powder Metall Inst. 2019;26(6):481-486. Published online December 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.6.481
- 1,352 View
- 9 Download
-
 Abstract
Abstract
 PDF
PDF Iron oxides currently attract considerable attention due to their potential applications in the fields of lithiumion batteries, bio-medical sensors, and hyperthermia therapy materials. Magnetite (Fe3O4) is a particularly interesting research target due to its low cost, good biocompatibility, outstanding stability in physiological conditions. Hydrothermal synthesis is one of several liquid-phase synthesis methods with water or an aqueous solution under high pressure and high temperature. This paper reports the growth of magnetic Fe3O4 particles from iron powder (spherical, <10 μm) through an alkaline hydrothermal process under the following conditions: (1) Different KOH molar concentrations and (2) different synthesis time for each KOH molar concentrations. The optimal condition for the synthesis of Fe3O4 using Fe powders is hydrothermal oxidation with 6.25 M KOH for 48 h, resulting in 89.2 emu/g of saturation magnetization at room temperature. The structure and morphologies of the synthesized particles are characterized by X-ray diffraction (XRD, 2θ = 20°–80°) with Cu-kα radiation and field emission scanning electron microscopy (FE-SEM), respectively. The magnetic properties of magnetite samples are investigated using a vibrating sample magnetometer (VSM). The role of KOH in the formation of magnetite octahedron is observed.
- [Korean]
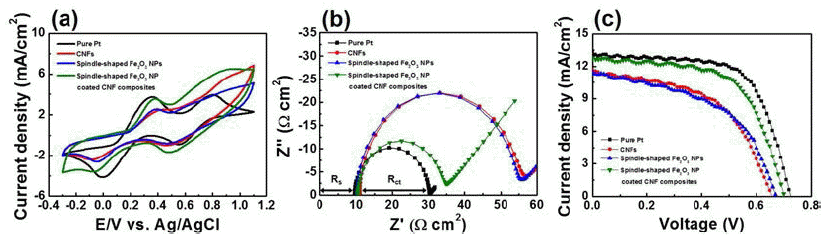
- Spindle-shaped Fe2O3 Nanoparticle Coated Carbon Nanofiber Composites for Low-cost Dye-sensitized Solar Cells
- Dong-Hyeun Oh, HyeLan An, Bon-Ryul Koo, Hyo-Jin Ahn
- J Korean Powder Metall Inst. 2016;23(2):95-101. Published online April 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.2.95
- 1,241 View
- 3 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF Carbon nanofiber (CNF) composites coated with spindle-shaped Fe2O3 nanoparticles (NPs) are fabricated by a combination of an electrospinning method and a hydrothermal method, and their morphological, structural, and chemical properties are measured by field-emission scanning electron microscopy, transmission electron microscopy, Xray diffraction, and X-ray photoelectron spectroscopy. For comparison, CNFs and spindle-shaped Fe2O3 NPs are prepared by either an electrospinning method or a hydrothermal method, respectively. Dye-sensitized solar cells (DSSCs) fabricated with the composites exhibit enhanced open circuit voltage (0.70 V), short-circuit current density (12.82 mA/cm2), fill factor (61.30%), and power conversion efficiency (5.52%) compared to those of the CNFs (0.66 V, 11.61 mA/cm2, 51.96%, and 3.97%) and spindle-shaped Fe2O3 NPs (0.67 V, 11.45 mA/cm2, 50.17%, and 3.86%). This performance improvement can be attributed to a synergistic effect of a superb catalytic reaction of spindle-shaped Fe2O3 NPs and efficient charge transfer relative to the one-dimensional nanostructure of the CNFs. Therefore, spindle-shaped Fe2O3-NPcoated CNF composites may be proposed as a potential alternative material for low-cost counter electrodes in DSSCs.
-
Citations
Citations to this article as recorded by- Ni Nanoparticles-Graphitic Carbon Nanofiber Composites for Pt-Free Counter Electrode in Dye-Sensitized Solar Cells
Dong-Hyeun Oh, Bon-Ryul Koo, Yu-Jin Lee, HyeLan An, Hyo-Jin Ahn
Korean Journal of Materials Research.2016; 26(11): 649. CrossRef
- Ni Nanoparticles-Graphitic Carbon Nanofiber Composites for Pt-Free Counter Electrode in Dye-Sensitized Solar Cells
TOP
 KPMI
KPMI


 First
First Prev
Prev


