Search
- Page Path
- HOME > Search
- [Korean]
- Mechanical Properties and Thermal Stability of Ti0.5Al0.5N/CrN Nano-multilayered Coatings
- Seung-Su Ahn, Jong-Keuk Park, Kyung-Sik Oh, Tai-Joo Chung
- J Korean Powder Metall Inst. 2020;27(5):406-413. Published online October 1, 2020
- DOI: https://doi.org/10.4150/KPMI.2020.27.5.406

- 664 View
- 2 Download
-
 Abstract
Abstract
 PDF
PDF Ti0.5Al0.5N/CrN nano-multilayers, which are known to exhibit excellent wear resistances, were prepared using the unbalanced magnetron sputter for various periods of 2–7 nm. Ti0.5Al0.5N and CrN comprised a cubic structure in a single layer with different lattice parameters; however, Ti0.5Al0.5N/CrN exhibited a cubic structure with the same lattice parameters that formed the superlattice in the nano-multilayers. The Ti0.5Al0.5/CrN multilayer with a period of 5.0 nm exceeded the hardness of the Ti0.5Al0.5N/CrN single layer, attaining a value of 36 GPa. According to the low-angle X-ray diffraction, the Ti0.5Al0.5N/CrN multilayer maintained its as-coated structure up to 700°C and exhibited a hardness of 32 GPa. The thickness of the oxidation layer of the Ti0.5Al0.5N/CrN multilayered coating was less than 25% of that of the single layers. Thus, the Ti0.5Al0.5N/CrN multilayered coating was superior in terms of hardness and oxidation resistance as compared to its constituent single layers.
- [Korean]
- Research Trends of the Mo-Si-B Alloys as Next Generation Ultra-high-temperature Alloys
- Won June Choi, Chun Woong Park, Jung Hyo Park, Young Do Kim, Jong Min Byun
- J Korean Powder Metall Inst. 2019;26(2):156-165. Published online April 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.2.156

- 1,973 View
- 27 Download
- 4 Citations
-
 Abstract
Abstract
 PDF
PDF Over the last decade, the next generation’s ultra-high-temperature materials as an alternative to Nickel-based superalloys have been highlighted. Ultra-high-temperature materials based on refractory metals are one of several potential candidates. In particular, molybdenum alloys with small amounts of silicon and boron (Mo-Si-B alloys) have superior properties at high temperature. However, research related to Mo-Si-B alloys were mainly conducted by several developed countries but garnered little interest in Korea. Therefore, in this review paper, we introduce the development history of Mo-Si-B alloys briefly and discuss the properties, particularly the mechanical and oxidation properties of Mo-Si-B alloys. We also introduce the latest research trends of Mo-Si-B alloys based on the research paper. Finally, for domestic research related to this field, we explain why Mo-Si-B alloys should be developed and suggest the potential directions for Mo-Si-B alloys research.
-
Citations
Citations to this article as recorded by- Thermal Stability and Weight Reduction of Al0.75V2.82CrZr Refractory High Entropy Alloy Prepared Via Mechanical Alloying
Minsu Kim, Hansung Lee, Byungmin Ahn
journal of Korean Powder Metallurgy Institute.2023; 30(6): 478. CrossRef - Preparation and Structure of Chromium Coatings Doped with Diamond Nanoparticles Deposited Directly on a Monolithic Composite of Molybdenum and Aluminum
V. P. Petkov, M. K. Aleksandrova, R. V. Valov, V. P. Korzhov, V. M. Kiiko, I. S. Zheltyakova
Protection of Metals and Physical Chemistry of Surfaces.2023; 59(3): 396. CrossRef - A Review of Mo-Si Intermetallic Compounds as Ultrahigh-Temperature Materials
Liang Jiang, Bin Zheng, Changsong Wu, Pengxiang Li, Tong Xue, Jiandong Wu, Fenglan Han, Yuhong Chen
Processes.2022; 10(9): 1772. CrossRef - Heat-Resistant Molybdenum Borosilicate Alloys Hardened with Titanium Carbides: Mo–Si–B–TiC (Survey)
I. L. Svetlov, O. G. Ospennikova, M. I. Karpov, Yu. V. Artemenko
Inorganic Materials: Applied Research.2021; 12(4): 866. CrossRef
- Thermal Stability and Weight Reduction of Al0.75V2.82CrZr Refractory High Entropy Alloy Prepared Via Mechanical Alloying
- [Korean]
- Lattice Deformation and Improvement Oxidation Resistance of Ti-6Al-4V Alloy Powders Prepared by Hydrogen Added Argon Heat Treatment
- Gye-Hoon Cho, Jung-Min Oh, Jae-Won Lim
- J Korean Powder Metall Inst. 2019;26(2):126-131. Published online April 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.2.126

- 704 View
- 1 Download
-
 Abstract
Abstract
 PDF
PDF In the present work, a new hydrogen added argon heat treatment process that prevents the formation of hydrides and eliminates the dehydrogenation step, is developed. Dissolved hydrogen has a good effect on sintering properties such as oxidation resistance and density of greens. This process can also reduce costs and processing time. In the experiment, commercially available Ti-6Al-4V powders are used. The powders are annealed using tube furnace in an argon atmosphere at 700°C and 900°C for 120 min. Hydrogen was injected temporarily during argon annealing to dissolve hydrogen, and a dehydrogenation process was performed simultaneously under an argon-only atmosphere. Without hydride formation, hydrogen was dissolved in the Ti-6Al-4V powder by X-ray diffraction and gas analysis. Hydrogen is first solubilized on the beta phase and expanded the beta phases’ cell volume. TGA analysis was carried out to evaluate the oxidation resistance, and it is confirmed that hydrogen-dissolved Ti-6Al-4V powders improves oxidation resistance more than raw materials.
- [Korean]
- The Effect of SiO2 addition on Oxidation and Electrical Resistance Stability at High-temperature of P/M Fecralloy Compact
- Jin-Woo Park, Jin-Uk Ok, Woo-young Jung, Dong-kyu Park, In-Shup Ahn
- J Korean Powder Metall Inst. 2017;24(4):292-297. Published online August 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.4.292
- 1,397 View
- 3 Download
-
 Abstract
Abstract
 PDF
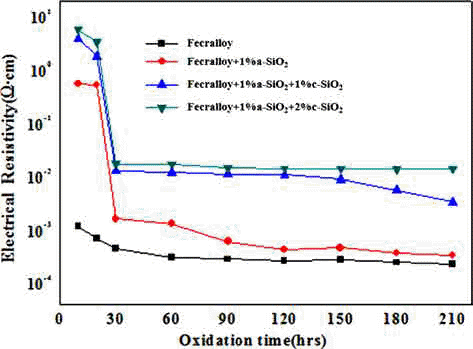
PDF A metallic oxide layer of a heat-resistant element contributes to the high-temperature oxidation resistance by delaying the oxidation and has a positive effect on the increase in electrical resistivity. In this study, green compacts of Fecralloy powder mixed with amorphous and crystalline silica are oxidized at 950°C for up to 210 h in order to evaluate the effect of metal oxide on the oxidation and electrical resistivity. The weight change ratio increases as per a parabolic law, and the increase is larger than that observed for Fecralloy owing to the formation of Fe-Si, Fe-Cr composite oxide, and Al2O3 upon the addition of Si oxide. Si oxides promote the formation of Al2O3 and Cr oxide at the grain boundary, and obstruct neck formation and the growth of Fecralloy particles to ensure stable electrical resistivity.
- [Korean]
- A Study on the Development of Compactability and Electrical Resistivity for P/M Fecralloy
- Jin-Woo Parka, Byung-Hyun Ko, Woo-Young Jung, Dong-Kyu Park, In-Shup Ahn
- J Korean Powder Metall Inst. 2016;23(6):426-431. Published online December 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.6.426
- 734 View
- 2 Download
-
 Abstract
Abstract
 PDF
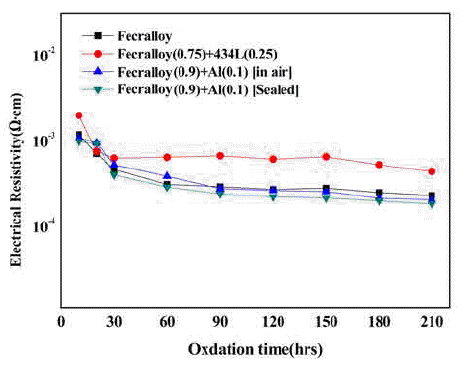
PDF The Fe-Cr-Al alloy system shows an excellent heat resistance because of the formation of an Al2O3 film on the metal surface in an oxidizing atmosphere at high temperatures up to 1400°C. The Fecralloy needs an additive that can act as a binder because of its bad compactability. In this study, the green compacts of STS434L and Al powder added to Fecralloy are oxidized at 950°C for up to 210 h. Fecralloy and Al is mixed by two types of ball milling. One is vented to air and the other was performed in a sealed jar. In the case of Al addition, there are no significant changes in the electrical resistance. Before the oxidation test, Al oxides are present in the Fecralloy surface, as determined from the energy dispersive spectroscopy results. The addition of Al improves the compactability because of an increased density, and the addition of STS434L increases the electrical resistivity by forming a composite oxide.
- [Korean]
- The Effect of Oxide Compound on Electrical Resistivity and Oxidation Stability in High-temperature for Ferritic P/M Stainless Steel
- Jin-Woo Park, Byung-Hyun Ko, Woo-young Jung, Dong-Kyu Park, In-Shup Ahn
- J Korean Powder Metall Inst. 2016;23(3):240-246. Published online June 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.3.240

- 758 View
- 1 Download
-
 Abstract
Abstract
 PDF
PDF In order to improve the high-temperature oxidation stability, sintered 434L stainless steel is studied, focusing on the effect of the addition of metallic oxides to form stable oxide films on the inner particle surface. The green compacts of Fecralloy powder or amorphous silica are added on STS434L and oxidized at 950°C up to 210 h. The weight change ratio of 434L with amorphous silica is higher than that of 434L mixed with Fecralloy, and the weight increase follows a parabolic law, which implies that the oxide film grows according to oxide diffusion through the densely formed oxide film. In the case of 434L mixed with Fecralloy, the elements in the matrix diffuse through the grain boundaries and form Al2O3 and Fe-Cr oxides. Stable high temperature corrosion resistance and electrical resistivity are obtained for STS434L mixed with Fecralloy.
TOP
 KPMI
KPMI


 First
First Prev
Prev


