Search
- Page Path
- HOME > Search
- [Korean]
- Cobalt Recovery by Oxalic Acid and Hydroxide Precipitation from Waste Cemented Carbide Scrap Cobalt Leaching Solution
- Jaesung Lee, Mingoo Kim, Seulgi Kim, Dongju Lee
- J Korean Powder Metall Inst. 2021;28(6):497-501. Published online December 1, 2021
- DOI: https://doi.org/10.4150/KPMI.2021.28.6.497
- 1,633 View
- 22 Download
-
 Abstract
Abstract
 PDF
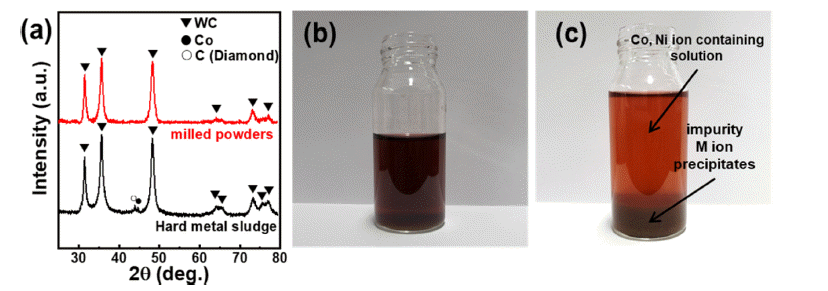
PDF Cobalt (Co) is mainly used to prepare cathode materials for lithium-ion batteries (LIBs) and binder metals for WC-Co hard metals. Developing an effective method for recovering Co from WC-Co waste sludge is of immense significance. In this study, Co is extracted from waste cemented carbide soft scrap via mechanochemical milling. The leaching ratio of Co reaches approximately 93%, and the leached solution, from which impurities except nickel are removed by pH titration, exhibits a purity of approximately 97%. The titrated aqueous Co salts are precipitated using oxalic acid and hydroxide precipitation, and the effects of the precipitating agent (oxalic acid and hydroxide) on the cobalt microstructure are investigated. It is confirmed that the type of Co compound and the crystal growth direction change according to the precipitation method, both of which affect the microstructure of the cobalt powders. This novel mechanochemical process is of significant importance for the recovery of Co from waste WC-Co hard metal. The recycled Co can be applied as a cemented carbide binder or a cathode material for lithium secondary batteries.
- [Korean]
- Current Status of Smelting and Recycling Technologies of Tungsten
- Ho-Sang Sohn
- J Korean Powder Metall Inst. 2021;28(4):342-351. Published online August 1, 2021
- DOI: https://doi.org/10.4150/KPMI.2021.28.4.342

- 2,308 View
- 58 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF Because of its unique properties, tungsten is a strategic and rare metal used in various industrial applications. However, the world's annual production of tungsten is only 84000 t. Ammonium paratungstate (APT), which is used as the main intermediate in industrial tungsten production, is usually obtained from tungsten concentrates of wolframite and scheelite by hydrometallurgical treatment. Intermediates such as tungsten trioxide, tungsten blue oxide, tungstic acid, and ammonium metatungstate can be derived from APT by thermal decomposition or chemical attack. Tungsten metal powder is produced through the hydrogen reduction of high-purity tungsten oxides, and tungsten carbide powder is produced by the reaction of tungsten powder and carbon black powder at 1300–1700°C in a hydrogen atmosphere. Tungsten scrap can be divided into hard and soft scrap based on shape (bulk or powder). It can also be divided into new scrap generated during the production of tungsten-bearing goods and old scrap collected at the end of life. Recycling technologies for tungsten can be divided into four main groups: direct, chemical, and semi-direct recycling, and melting metallurgy. In this review, the current status of tungsten smelting and recycling technologies is discussed.
-
Citations
Citations to this article as recorded by- Synthesis and thermal properties of single-phase AMT crystals via alcoholic precipitation
Haoyu Liu, Wendi Zhang, Qiusheng Zhou, Xiaobin Li, Jie Li, Guihua Liu, Zhihong Peng, Tiangui Qi, Leiting Shen, Yilin Wang
Canadian Metallurgical Quarterly.2026; 65(2): 2088. CrossRef - The Current Status and Securing Strategies of Core Mineral Tungsten Resources
Dohyun Jeong, Seongmin Kim, Hoseok Jeon
Journal of the Korean Society of Mineral and Energy Resources Engineers.2023; 60(5): 341. CrossRef - Tungsten distribution and vertical migration in soils near a typical abandoned tungsten smelter
Huihui Du, Yang Li, Dan Wan, Chuanqiang Sun, Jing Sun
Journal of Hazardous Materials.2022; 429: 128292. CrossRef
- Synthesis and thermal properties of single-phase AMT crystals via alcoholic precipitation
- [Korean]
- Property Evaluation of Tungsten-Carbide Hard Materials as a Function of Binder
- Ju-Hun Kim, Ik-Hyun Oh, Jeong-Han Lee, Sung-Kil Hong, Hyun-Kuk Park
- J Korean Powder Metall Inst. 2019;26(2):132-137. Published online April 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.2.132
- 1,316 View
- 8 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
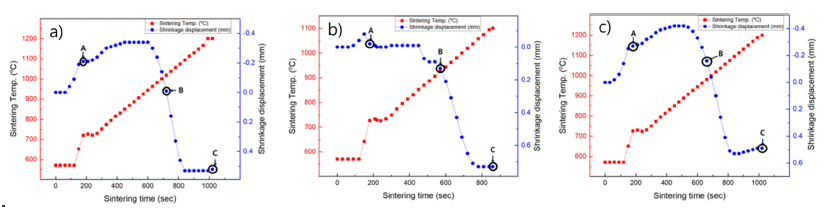
PDF Tungsten carbide (WC) hard materials are used in various industries and possess a superior hardness compared to other hard materials. They have particularly high melting points, high strength, and abrasion resistance. Accordingly, tungsten carbide hard materials are used for wear-resistant tools, cutting tools, machining tools, and other tooling materials. In this study, the WC-5wt.%Co, Fe, Ni hard materials are densified using the horizontal ball milled WC-Co, WC-Fe, and WC-Ni powders by a spark plasma sintering process. The WC-5Co, WC-5Fe, and WC-5Ni hard materials are almost completely densified with a relative density of up to 99.6% after simultaneous application of a pressure of 60 MPa and an electric current for about 15 min without any significant change in the grain size. The average grain size of WC-5Co, WC-5Fe, and WC-5Ni that was produced through SPS was about 0.421, 0.779, and 0.429 μm, respectively. The hardness and fracture toughness of the dense WC-5Co, WC-5Fe, WC-5Ni hard materials were also investigated.
-
Citations
Citations to this article as recorded by- Enhancing Mechanical Properties via Grain Growth Suppression and High Densification in WC Compacts
Jong Min Gwak, Min Soo Park, Gook Hyun Ha, Nam Hyun Kang
Metals and Materials International.2025; 31(12): 3733. CrossRef - Synthesis of W2C by Spark Plasma Sintering of W-WC Powder Mixture and Its Etching Property
Gyu-Sang Oh, Sung-Min Lee, Sung-Soo Ryu
Journal of Korean Powder Metallurgy Institute.2020; 27(4): 293. CrossRef - Fabrication and Properties of Densified Tungsten by Magnetic Pulse Compaction and Spark Plasma Sintering
Eui Seon Lee, Jongmin Byun, Young-Keun Jeong, Sung-Tag Oh
Korean Journal of Materials Research.2020; 30(6): 321. CrossRef
- Enhancing Mechanical Properties via Grain Growth Suppression and High Densification in WC Compacts
- [English]
- Coating of Cobalt Over Tungsten Carbide Powder by Wet Chemical Reduction Method
- Hyun-Seon Hong, Jin-Ho Yoon
- J Korean Powder Metall Inst. 2014;21(2):93-96. Published online April 1, 2014
- DOI: https://doi.org/10.4150/KPMI.2014.21.2.93
- 2,202 View
- 9 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
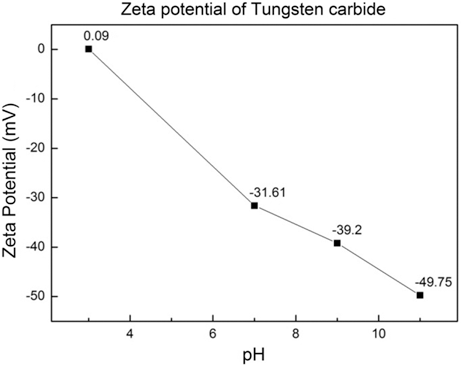
PDF Cobalt coated tungsten carbide-cobalt composite powder has been prepared through wet chemical reduction method. The cobalt sulfate solution was converted to the cobalt chloride then the cobalt hydroxide. The tungsten carbide powders were added in to the cobalt hydroxide, the cobalt hydroxide was reduced and coated over tungsten carbide powder using hypo-phosphorous acid. Both the cobalt and the tungsten carbide phase peaks were evident in the tungsten carbide-cobalt composite powder by X-ray diffraction. The average particle size measured via scanning electron microscope, particle size analysis was around 380 nm and the thickness of coated cobalt was determined to be 30~40 nm by transmission electron microscopy.
-
Citations
Citations to this article as recorded by- Electroless Ni-P deposition on WC powders through direct PdCl2 activation and study on the underlying mechanisms
Peng Tang, Shuwen Jiang, Jiawei Yan, Xianquan Li
Next Materials.2025; 6: 100496. CrossRef - Pre-treatments of initial materials for controlling synthesized TaC characteristics in the SHS process
Jae Jin Sim, Sang Hoon Choi, Ji Hwan Park, Il Kyu Park, Jae Hong Lim, Kyoung Tae Park
journal of Korean Powder Metallurgy Institute.2018; 25(3): 251. CrossRef - Spark plasma sintering of WC–Co tool materials prepared with emphasis on WC core–Co shell structure development
Sungkyu Lee, Hyun Seon Hong, Hyo-Seob Kim, Soon-Jik Hong, Jin-Ho Yoon
International Journal of Refractory Metals and Hard Materials.2015; 53: 41. CrossRef
- Electroless Ni-P deposition on WC powders through direct PdCl2 activation and study on the underlying mechanisms
TOP
 KPMI
KPMI


 First
First Prev
Prev


