Search
- Page Path
- HOME > Search
- [Korean]
- A Study on Morphology Control of (Ga1-xZnx)(N1-xOx) Nanofibers according to the Composition and Crystallinity of Oxide Nanofibers Synthesized by Electrospinning
- Jeong Hyun Kim, Sung-Tag Oh, Young-In Lee
- J Korean Powder Metall Inst. 2021;28(3):259-266. Published online June 1, 2021
- DOI: https://doi.org/10.4150/KPMI.2021.28.3.259

- 779 View
- 2 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF The (Ga1-xZnx)(N1-xOx) solid solution is attracting extensive attention for photocatalytic water splitting and wastewater treatment owing to its narrow and controllable band gap. To optimize the photocatalytic performance of the solid solution, the key points are to decrease its band gap and recombination rate. In this study, (Ga1-xZnx)(N1-xOx) nanofibers with various Zn fractions are prepared by electrospinning followed by calcination and nitridation. The effect of the composition and crystallinity of electrospun oxide nanofibers on the morphology and optical properties of the obtained solid-solution nanofibers are systematically investigated. The results show that the final shape of the (Ga1-xZnx) (N1-xOx) material is greatly affected by the crystallinity of the oxide nanofibers before nitridation. The photocatalytic properties of (Ga1-xZnx)(N1-xOx) with different Ga:Zn atomic ratios are investigated by studying the degradation of rhodamine B under visible light irradiation.
-
Citations
Citations to this article as recorded by- Fabrication of Nanowire by Electrospinning Process Using Nickel Oxide Particle Recovered from MLCC
Haein Shin, Jongwon Bae, Minsu Kang, Kun-Jae Lee
journal of Korean Powder Metallurgy Institute.2023; 30(6): 502. CrossRef
- Fabrication of Nanowire by Electrospinning Process Using Nickel Oxide Particle Recovered from MLCC
- [Korean]
- Synthesis and Optical Property of (GaN)1-x(ZnO)x Nanoparticles Using an Ultrasonic Spray Pyrolysis Process and Subsequent Chemical Transformation
- Jeong Hyun Kim, Cheol-Hui Ryu, Myungjun Ji, Yomin Choi, Young-In Lee
- J Korean Powder Metall Inst. 2021;28(2):143-149. Published online April 1, 2021
- DOI: https://doi.org/10.4150/KPMI.2021.28.2.143
- 930 View
- 2 Download
-
 Abstract
Abstract
 PDF
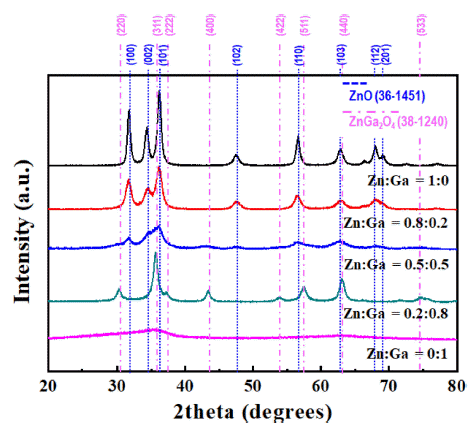
PDF In this study, (GaN)1-x(ZnO)x solid solution nanoparticles with a high zinc content are prepared by ultrasonic spray pyrolysis and subsequent nitridation. The structure and morphology of the samples are investigated by X-ray diffraction (XRD), field-emission scanning electron microscopy, and energy-dispersive X-ray spectroscopy. The characterization results show a phase transition from the Zn and Ga-based oxides (ZnO or ZnGa2O4) to a (GaN)1-x (ZnO)x solid solution under an NH3 atmosphere. The effect of the precursor solution concentration and nitridation temperature on the final products are systematically investigated to obtain (GaN)1-x(ZnO)x nanoparticles with a high Zn concentration. It is confirmed that the powder synthesized from the solution in which the ratio of Zn and Ga was set to 0.8:0.2, as the initial precursor composition was composed of about 0.8-mole fraction of Zn, similar to the initially set one, through nitriding treatment at 700°C. Besides, the synthesized nanoparticles exhibited the typical XRD pattern of (GaN)1-x(ZnO)x, and a strong absorption of visible light with a bandgap energy of approximately 2.78 eV, confirming their potential use as a hydrogen production photocatalyst.
- [Korean]
- Effect of Calcination Temperature on the Microstructure and Photocatalytic Activity of Electrospun BiVO4 Nanofiber
- Myeongjun Ji, Jeong Hyun Kim, Cheol-Hui Ryu, Yun Taek Ko, Young-In Lee
- J Korean Powder Metall Inst. 2020;27(3):226-232. Published online June 1, 2020
- DOI: https://doi.org/10.4150/KPMI.2020.27.3.226

- 1,406 View
- 5 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF Bismuth vanadate (BiVO4) is considered a potentially attractive candidate for the visible-light-driven photodegradation of organic pollutants. In an effort to enhance their photocatalytic activities, BiVO4 nanofibers with controlled microstructures, grain sizes, and crystallinities are successfully prepared by electrospinning followed by a precisely controlled heat treatment. The structural features, morphologies, and photo-absorption performances of the asprepared samples are systematically investigated and can be readily controlled by varying the calcination temperature. From the physicochemical analysis results of the synthesized nanofiber, it is found that the nanofiber calcines at a lower temperature, shows a smaller crystallite size, and lower crystallinity. The photocatalytic degradation of rhodamine-B (RhB) reveals that the photocatalytic activity of the BiVO4 nanofibers can be improved by a thermal treatment at a relatively low temperature because of the optimization of the conflicting characteristics, crystallinity, crystallite size, and microstructure. The photocatalytic activity of the nanofiber calcined at 350°C for the degradation of RhB under visible-light irradiation exhibits a greater photocatalytic activity than the nanofibers synthesized at 400°C and 450°C.
-
Citations
Citations to this article as recorded by- Design, synthesis, and characterization of a porous ceramic-supported CeO2 nanocatalyst for CO -free H2 evolution
Jimin Lee, Minseob Lim, Tae Sung Kim, Kee-Ryung Park, Jong-Sik Lee, Hong-Baek Cho, Joo Hyun Park, Yong-Ho Choa
Applied Surface Science.2021; 548: 149198. CrossRef
- Design, synthesis, and characterization of a porous ceramic-supported CeO2 nanocatalyst for CO -free H2 evolution
- [Korean]
- Effect of Pyrolysis temperature on TiO2 Nanoparticles Synthesized by a Salt-assisted Ultrasonic Spray Pyrolysis Process
- Jae-Hyun Yoo, Myeong-Jun Ji, Woo-Young Park, Young-In Lee
- J Korean Powder Metall Inst. 2019;26(3):237-242. Published online June 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.3.237

- 1,048 View
- 6 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF In this study, ultrasonic spray pyrolysis combined with salt-assisted decomposition, a process that adds sodium nitrate (NaNO3) into a titanium precursor solution, is used to synthesize nanosized titanium dioxide (TiO2) particles. The added NaNO3 prevents the agglomeration of the primary nanoparticles in the pyrolysis process. The nanoparticles are obtained after a washing process, removing NaNO3 and NaF from the secondary particles, which consist of the salts and TiO2 nanoparticles. The effects of pyrolysis temperature on the size, crystallographic characteristics, and bandgap energy of the synthesized nanoparticles are systematically investigated. The synthesized TiO2 nanoparticles have a size of approximately 2–10 nm a bandgap energy of 3.1–3.25 eV, depending on the synthetic temperature. These differences in properties affect the photocatalytic activities of the synthesized TiO2 nanoparticles.
-
Citations
Citations to this article as recorded by- Microstructure and Sintering Behavior of Fine Tungsten Powders Synthesized by Ultrasonic Spray Pyrolysis
Hyeonhui Jo, Jeong Hyun Kim, Young-In Lee, Young-Keun Jeong, Sung-Tag Oh
Korean Journal of Metals and Materials.2021; 59(5): 289. CrossRef
- Microstructure and Sintering Behavior of Fine Tungsten Powders Synthesized by Ultrasonic Spray Pyrolysis
- [Korean]
- Synthesis and Optical Property of TiO2 Nanoparticles Using a Salt-assisted Ultrasonic Spray Pyrolysis Process
- Myeong-Jun Ji, Woo-Young Park, Jae-Hyun Yoo, Young-In Lee
- J Korean Powder Metall Inst. 2019;26(1):34-39. Published online February 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.1.34
- 1,006 View
- 1 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF Current synthesis processes for titanium dioxide (TiO2) nanoparticles require expensive precursors or templates as well as complex steps and long reaction times. In addition, these processes produce highly agglomerated nanoparticles. In this study, we demonstrate a simple and continuous approach to synthesize TiO2 nanoparticles by a salt-assisted ultrasonic spray pyrolysis method. We also investigate the effect of salt content in a precursor solution on the morphology and size of synthesized products. The synthesized TiO2 nanoparticles are systematically characterized by X-ray diffraction, transmission electron micrograph, and UV-Vis spectroscopy. These nanoparticles appear to have a single anatase phase and a uniform particle-size distribution with an average particle size of approximately 10 nm. By extrapolating the plots of the transformed Kubelka-Munk function versus the absorbed light energy, we determine that the energy band gap of the synthesized TiO2 nanoparticles is 3.25 eV.
-
Citations
Citations to this article as recorded by- Effect of Pyrolysis temperature on TiO2 Nanoparticles Synthesized by a Salt-assisted Ultrasonic Spray Pyrolysis Process
Jae-Hyun Yoo, Myeong-Jun Ji, Woo-Young Park, Young-In Lee
Journal of Korean Powder Metallurgy Institute.2019; 26(3): 237. CrossRef
- Effect of Pyrolysis temperature on TiO2 Nanoparticles Synthesized by a Salt-assisted Ultrasonic Spray Pyrolysis Process
- [Korean]
- Synthesis and Optical Property of GaN Powder Using an Ultrasonic Spray Pyrolysis Process and Subsequent Nitridation Treatment
- Myeong-Jun Ji, Jae-Hyun Yoo, Young-In Lee
- J Korean Powder Metall Inst. 2018;25(6):482-486. Published online December 1, 2018
- DOI: https://doi.org/10.4150/KPMI.2018.25.6.482

- 1,052 View
- 6 Download
-
 Abstract
Abstract
 PDF
PDF Despite numerous advances in the preparation and use of GaN, and many leading-edge applications in lighting technologies, the preparation of high-quality GaN powder remains a challenge. Ammonolytic preparations of polycrystalline GaN have been studied using various precursors, but all were time-consuming and required high temperatures. In this study, an efficient and low-temperature method to synthesize high-purity hexagonal GaN powder is developed using sub-micron Ga2O3 powder as a starting material. The sub-micron Ga2O3 powder was prepared by an ultrasonic spray pyrolysis process. The GaN powder is synthesized from the sub-micron Ga2O3 powder through a nitridation treatment in an NH3 flow at 800°C. The characteristics of the synthesized powder are systematically examined by X-ray diffraction, scanning and transmission electron microscopy, and UV-vis spectrophotometer.
- [Korean]
- Synthesis and Optical Property of BaTiO3 Nanoparticles Using a Salt-assisted Ultrasonic Spray Pyrolysis Process
- Young Hwangbo, Young-In Lee
- J Korean Powder Metall Inst. 2017;24(4):326-331. Published online August 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.4.326
- 1,060 View
- 6 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF The structural formation of inorganic nanoparticles dispersed in polymer matrices is a key technology for producing advanced nanocomposites with a unique combination of optical, electrical, and mechanical properties. Barium titanate (BaTiO3) nanoparticles are attractive for increasing the refractive index and dielectric constant of polymer nanocomposites. Current synthesis processes for BaTiO3 nanoparticles require expensive precursors or organic solvents, complicated steps, and long reaction times. In this study, we demonstrate a simple and continuous approach for synthesizing BaTiO3 nanoparticles based on a salt-assisted ultrasonic spray pyrolysis method. This process allows the synthesis of BaTiO3 nanoparticles with diameters of 20-50 nm and a highly crystalline tetragonal structure. The optical properties and photocatalytic activities of the nanoparticles show that they are suitable for use as fillers in various nanocomposites.
-
Citations
Citations to this article as recorded by- Sr doping effects on La(1-x)SrxMnO3-BaTiO3 nanocomposites: A comprehensive analysis of structural, optical, magnetic, and dielectric properties
Milad Karamzadeh-Jahromi, Morteza Izadifard, Mohammad Ebrahim Ghazi
Journal of Alloys and Compounds.2024; 1006: 176272. CrossRef
- Sr doping effects on La(1-x)SrxMnO3-BaTiO3 nanocomposites: A comprehensive analysis of structural, optical, magnetic, and dielectric properties
- [Korean]
- Investigation on Microstructure and Electrical Properties of Silver Conductive Features Using a Powder Composed of Silver nanoparticles and Nanoplatelets
- Yong-Sung Goo, Yong-Ho Choa, Young Hwangbo, Young-In Lee
- J Korean Powder Metall Inst. 2016;23(5):358-363. Published online October 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.5.358
- 695 View
- 4 Download
-
 Abstract
Abstract
 PDF
PDF Noncontact direct-printed conductive silver patterns with an enhanced electrical resistivity are fabricated using a silver ink with a mixture of silver nanoparticles and nanoplates. The microstructure and electrical resistivity of the silver pattern are systematically investigated as a function of the mixing ratio of the nanoparticles and nanoplates. The pattern, which is fabricated using a mixture with a mixing ratio of 3(nanoparticles):7(nanoplates) and sintered at 200°C shows a highly dense and well-sintered microstructure and has a resistivity of 7.60 μΩ·cm. This originates a mutual synergistic effect through a combination of the sinterability of the nanoparticles and the packing ability of the nanoplates. This is a conductive material that can be used to fabricate noncontact direct-printed conductive patterns with excellent electrical conductivity for various flexible electronics applications, including solar cells, displays, RFIDs, and sensors.
- [Korean]
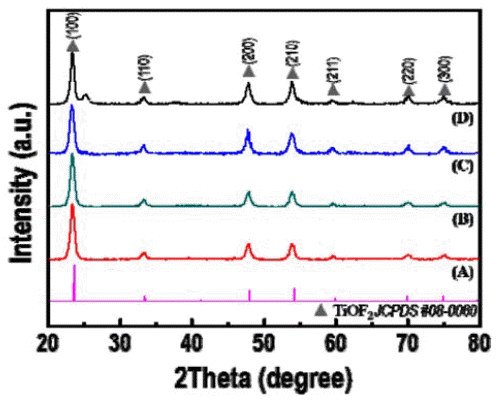
- Synthesis and Optical Property of a TiOF2 Powder via an Ultrasonic Spray Pyrolysis Process
- Young Hwangbo, Young-In Lee
- J Korean Powder Metall Inst. 2016;23(4):307-310. Published online August 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.4.307
- 866 View
- 3 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF TiOF2, which has remarkable electrochemical and optical properties, is used in various applications such as Li-ion batteries, electrochemical displays, and photocatalysts. In addition, it is possible to utilize the template which is allowed to synthesize fluorine doped TiO2 powders with hollow or faceted structures. However, common synthesis methods of TiOF2 powders have some disadvantages such as the use of expensive and harmful precursors and batchtype processes with a limited production scale. In this study, we report a synthetic route for preparing TiOF2 powders by using an inexpensive and harmless precursor and a continuous ultrasonic spray pyrolysis process under a controlled atmosphere to address the aforementioned problems. The synthesized powder has an average size of 1 μm, a spherical shape, a pure TiOF2 phase, and exhibits a band-gap energy of 3.2 eV.
-
Citations
Citations to this article as recorded by- Photocatalytic activity of rutile TiO2 powders coupled with anatase TiO2 nanoparticles using surfactant
Jong Min Byun, Chun Woong Park, Young In Kim, Young Do Kim
journal of Korean Powder Metallurgy Institute.2018; 25(3): 257. CrossRef
- Photocatalytic activity of rutile TiO2 powders coupled with anatase TiO2 nanoparticles using surfactant
- [Korean]
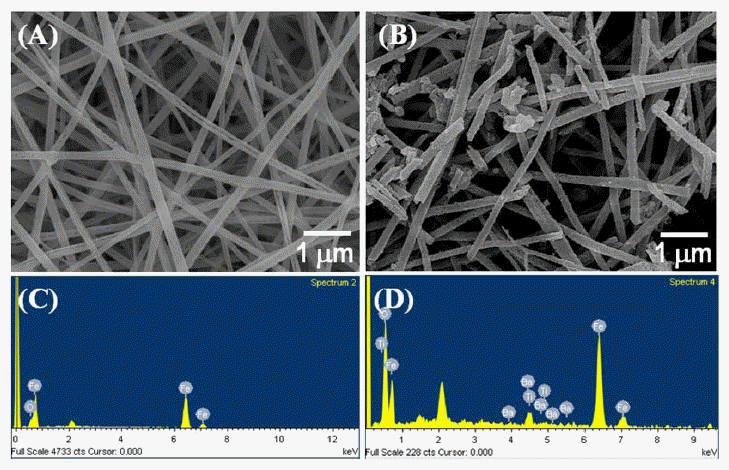
- Synthesis and Electromagnetic Wave Absorbing Property of BaTiO3@Fe Nanofibers with Core-Shell Structure
- Young-In Lee, Dae-Hwan Jang, Ki-Hoon Sung, Kyuman Lee, Yong-Ho Choa
- J Korean Powder Metall Inst. 2016;23(1):38-42. Published online February 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.1.38
- 1,188 View
- 7 Download
- 4 Citations
-
 Abstract
Abstract
 PDF
PDF BaTiO3-coated Fe nanofibers are synthesized via a three-step process. α-Fe2O3 nanofibers with an average diameter of approximately 200 nm are first prepared using an electrospinning process followed by a calcination step. The BaTiO3 coating layer on the nanofiber is formed by a sol-gel process, and a thermal reduction process is then applied to the core-shell nanofiber to selectively reduce the α-Fe2O3 to Fe. The thickness of the BaTiO3 shell is controlled by varying the reaction time. To evaluate the electromagnetic (EM) wave-absorbing abilities of the BaTiO3@Fe nanofiber, epoxy-based composites containing the nanofibers are fabricated. The composites show excellent EM wave absorption properties where the power loss increases to the high frequency region without any degradation. Our results demonstrate that the BaTiO3@Fe nanofibers obtained in this work are attractive candidates for electromagnetic wave absorption applications.
-
Citations
Citations to this article as recorded by- Research on Electromagnetic Wave Absorption Based on Electrospinning Technology†
Baoding Li, Jing Qiao, Yue Liu, Haoyuan Tian, Wei Liu, Qilei Wu, Zhou Wang, Jiurong Liu, Zhihui Zeng
Chinese Journal of Chemistry.2024; 42(7): 777. CrossRef - Design, synthesis, and characterization of a porous ceramic-supported CeO2 nanocatalyst for CO -free H2 evolution
Jimin Lee, Minseob Lim, Tae Sung Kim, Kee-Ryung Park, Jong-Sik Lee, Hong-Baek Cho, Joo Hyun Park, Yong-Ho Choa
Applied Surface Science.2021; 548: 149198. CrossRef - Electromagnetic wave absorption properties of Fe/MgO composites synthesized by a simple ultrasonic spray pyrolysis method
Hyo-Ryoung Lim, Seung-Jae Jung, Tae-Yeon Hwang, Jimin Lee, Ki Hyeon Kim, Hong-baek Cho, Yong-Ho Choa
Applied Surface Science.2019; 473: 1009. CrossRef - Study on the Optimization of Reduction Conditions for Samarium-Cobalt Nanofiber Preparation
Jimin Lee, Jongryoul Kim, Yong-Ho Choa
Journal of Korean Powder Metallurgy Institute.2019; 26(4): 334. CrossRef
- Research on Electromagnetic Wave Absorption Based on Electrospinning Technology†
- [Korean]
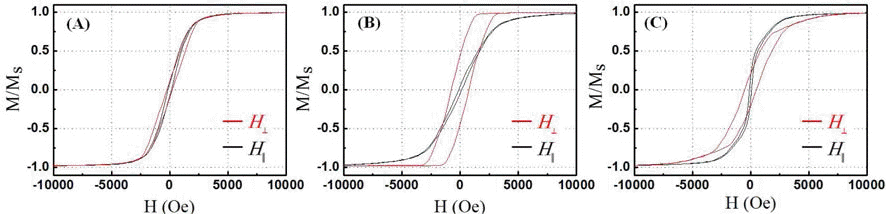
- Synthesis and Characterization of Nickel Nanowires by an Anodic Aluminum Oxide Template-Based Electrodeposition
- Hyo-Ryoung Lim, Yong-Ho Choa, Young-In Lee
- J Korean Powder Metall Inst. 2015;22(3):216-220. Published online June 1, 2015
- DOI: https://doi.org/10.4150/KPMI.2015.22.3.216
- 930 View
- 2 Download
-
 Abstract
Abstract
 PDF
PDF Vertically oriented nickel nanowire arrays with a different diameter and length are synthesized in porous anodic aluminium oxide templates by an electrodeposition method. The pore diameters of the templates are adjusted by controlling the anodization conditions and then they are utilized as templates to grow nickel nanowire arrays. The nickel nanowires have the average diameters of approximately 25 and 260 nm and the crystal structure, morphology and microstructure of the nanowires are systematically investigated using XRD, FE-SEM and TEM analysis. The nickel nanowire arrays show a magnetic anisotropy with the easy axis parallel to the nanowires and the coercivity and remanence enhance with decreasing a wire diameter and increasing a wire length.
- [Korean]
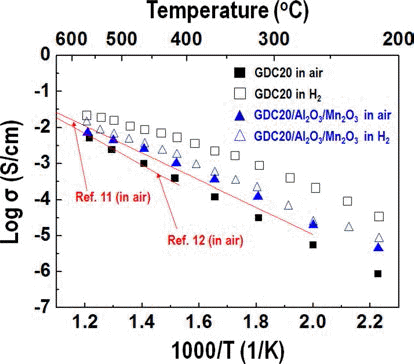
- Synthesis and Characterization of a Ceria Based Composite Electrolyte for Solid Oxide Fuel Cells by an Ultrasonic Spray Pyrolysis Process
- Young-In Lee, Yong-Ho Choa
- J Korean Powder Metall Inst. 2014;21(3):222-228. Published online June 1, 2014
- DOI: https://doi.org/10.4150/KPMI.2014.21.3.222
- 1,059 View
- 2 Download
- 2 Citations
-
 Abstract
Abstract
 PDF
PDF Much research into fuel cells operating at a temperature below 800°C. is being performed. There are significant efforts to replace the yttria-stabilized zirconia electrolyte with a doped ceria electrolyte that has high ionic conductivity even at a lower temperature. Even if the doped ceria electrolyte has high ionic conductivity, it also shows high electronic conductivity in a reducing environment, therefore, when used as a solid electrolyte of a fuel cell, the powergeneration efficiency and mechanical properties of the fuel cell may be degraded. In this study, gadolinium-doped ceria nanopowder with Al2O3 and Mn2O3 as a reinforcing and electron trapping agents were synthesized by ultrasonic pyrolysis process. After firing, their microstructure and mechanical and electrical properties were investigated and compared with those of pure gadolinium-doped ceria specimen.
-
Citations
Citations to this article as recorded by- High growth-rate atomic layer deposition process of cerium oxide thin film for solid oxide fuel cell
Jin-Geun Yu, Byung Chan Yang, Jeong Woo Shin, Sungje Lee, Seongkook Oh, Jae-Ho Choi, Jaehack Jeong, Wontae Noh, Jihwan An
Ceramics International.2019; 45(3): 3811. CrossRef - Atomic-layer-deposited ZrO2-doped CeO2 thin film for facilitating oxygen reduction reaction in solid oxide fuel cell
Byung Chan Yang, Dohyun Go, Seongkook Oh, Jeong Woo Shin, Hyong June Kim, Jihwan An
Applied Surface Science.2019; 473: 102. CrossRef
- High growth-rate atomic layer deposition process of cerium oxide thin film for solid oxide fuel cell
- [Korean]
- Synthesis and Characterization of Cu Nanowires Using Anodic Alumina Template Based Electrochemical Deposition Method
- Young-In Lee, Yong-Ho Choa
- J Korean Powder Metall Inst. 2012;19(5):367-372.
- DOI: https://doi.org/10.4150/KPMI.2012.19.5.367

- 659 View
- 1 Download
-
 Abstract
Abstract
 PDF
PDF - Single crystalline Cu nanowires with controlled diameters and aspect ratios have been synthesized using electrochemical deposition within confined nanochannels of a porous anodic aluminium oxide(AAO) template. The diameters of nano-sized cylindrical pores in AAO template were adjusted by controlling the anodization conditions. Cu nanowires with diameters of approximately 38, 99, 274 nm were synthesized by the electrodeposition using the AAO templates. The crystal structure, morphology and microstructure of the Cu nanowires were systematically investigated using XRD, FE-SEM, TEM and SAED. Investigation results revealed that the Cu nanowires had the controlled diameter, high aspect ratio and single crystalline nature.
- [English]
- Low Temperature Debinding Process Using Oxygen Plasma for Flexible Printed Electronics
- Young-In Lee
- J Korean Powder Metall Inst. 2012;19(5):343-347.
- DOI: https://doi.org/10.4150/KPMI.2012.19.5.343

- 1,014 View
- 1 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF - In this study, an oxygen plasma treatment was used as a low temperature debinding method to form a conductive copper feature on a flexible substrate using a direct printing process. To demonstrate this concept, conductive copper patterns were formed on polyimide films using a copper nanoparticle-based paste with polymeric binders and dispersing agents and a screen printing method. Thermal and oxygen plasma treatments were utilized to remove the polymeric vehicle before a sintering of copper nanoparticles. The effect of the debinding methods on the phase, microstructure and electrical conductivity of the screen-printed patterns was systematically investigated by FE-SEM, TGA, XRD and four-point probe analysis. The patterns formed using oxygen plasma debinding showed the well-developed microstructure and the superior electrical conductivity compared with those of using thermal debinding.
-
Citations
Citations to this article as recorded by- Research on new ceramic debinding technology with low energy consumption via dielectric barrier discharge
Yimeng Huang, Yong Liu, Nianping Yan, Xuetong Zhao, Xinhao Zhao, Yueji Li, Xilin Wang, Zhidong Jia
Physica Scripta.2023; 98(4): 045904. CrossRef - Green Manufacturing of Highly Conductive Cu2O and Cu Nanoparticles for Photonic-Sintered Printed Electronics
Young-Tae Kwon, Seung-Jae Yune, Yoseb Song, Woon-Hong Yeo, Yong-Ho Choa
ACS Applied Electronic Materials.2019; 1(10): 2069. CrossRef - Fabrication of Micro Pattern on Flexible Substrate by Nano Ink using Superhydrophobic Effect
Soo-Jung Son, Young-Sang Cho, Jong Joo Rha, Chul-Jin Cho
Journal of Korean Powder Metallurgy Institute.2013; 20(2): 120. CrossRef
- Research on new ceramic debinding technology with low energy consumption via dielectric barrier discharge
- [Korean]
- Synthesis of SnO2 Nanotubes Via Electrospinning Process and Their Application to Lithium Ion Battery Anodes
- Young-In Lee, Yong-Ho Choa
- J Korean Powder Metall Inst. 2012;19(4):271-277.
- DOI: https://doi.org/10.4150/KPMI.2012.19.4.271

- 1,062 View
- 0 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF - SnO_2 nanotubes were successfully synthesized using an electrospinning technique followed by calcination in air. The nanotubes were the single phase nature of SnO_2 and consisted of approximately 14 nm nanocrystals. SEM and TEM characterizations demonstrated that uniform hollow fibers with an average outer diameter of around 124 nm and wall thickness of around 25 nm were successfully obtained. As anode materials for lithium ion batteries, the SnO_2 nanotubes exhibited excellent cyclability and reversible capacity of 580mAhg-1 up to 25 cycles at 100mAg-1 as compared to SnO_2 nanoparticles with a capacity of sim200mAhg-1. Such excellent performance of the SnO_2 nanotube was related to the one-dimensional hollow structure which acted as a buffer zone during the volume contraction and expansion of Sn.
-
Citations
Citations to this article as recorded by- Fabrication of Nanowire by Electrospinning Process Using Nickel Oxide Particle Recovered from MLCC
Haein Shin, Jongwon Bae, Minsu Kang, Kun-Jae Lee
journal of Korean Powder Metallurgy Institute.2023; 30(6): 502. CrossRef - A study on the synthesis of tin oxide crystalline by the liquid reduction precipitation method and hydrothermal process
Il-Jeong Park, Geon-Hong Kim, Dae-Weon Kim, Hee-Lack Choi, Hang-Chul Jung
Journal of the Korean Crystal Growth and Crystal Technology.2016; 26(3): 95. CrossRef - Improvement of Triboelectric Efficiency using SnO2 Friction Layer for Triboelectric Generator
No Ho Lee, Jae Rok Shin, Ji Een Yoo, Dong Hun You, Bon-Ryul Koo, Sung Woo Lee, Hyo-Jin Ahn, Byung Joon Choi
Journal of Korean Powder Metallurgy Institute.2015; 22(5): 321. CrossRef
- Fabrication of Nanowire by Electrospinning Process Using Nickel Oxide Particle Recovered from MLCC
- [Korean]
- Microstructure and Mechanical Properties of Al2O3/Fe-Ni Nanocomposite Prepared by Rapid Sintering
- Young-In Lee, Kun-Jae Lee, Dae-Hwan Jang, Jae-Kyo Yang, Yong-Ho Cho
- J Korean Powder Metall Inst. 2010;17(3):203-208.
- DOI: https://doi.org/10.4150/KPMI.2010.17.3.203

- 705 View
- 1 Download
-
 Abstract
Abstract
 PDF
PDF - A new High Frequency Induction Heating (HFIH) process has been developed to fabricate dense Al_2O_3 reinforced with Fe-Ni magnetic metal dispersion particles. The process is based on the reduction of metal oxide particles immediately prior to sintering. The synthesized Al_2O_3/Fe-Ni nanocomposite powders were formed directly from the selective reduction of metal oxide powders, such as NiO and Fe_2O_3. Dense Al_2O_3/Fe-Ni nanocomposite was fabricated using the HFIH method with an extremely high heating rate of 2000°C/min. Phase identification and microstructure of nanocomposite powders and sintered specimens were determined by X-ray diffraction and SEM and TEM, respectively. Vickers hardness experiment were performed to investigate the mechanical properties of the Al_2O_3/Fe-Ni nanocomposite.
- [Korean]
- Research on Synthesis and Sintering Behavior of Nano-sized (Pb, La)TiO3 Powders Using Mechano Chemical Process
- Young-In Lee, Yong-Sung Goo, Jong-Sik Lee, Yong-Ho Choa
- J Korean Powder Metall Inst. 2010;17(2):101-106.
- DOI: https://doi.org/10.4150/KPMI.2010.17.2.101

- 790 View
- 2 Download
-
 Abstract
Abstract
 PDF
PDF - In this study, we successfully synthesized a nano-sized lanthanum-modified lead-titanate (PLT) powder with a perovskite structure using a high-energy mechanochemical process (MCP). In addition, the sintering behavior of synthesized PLT nanopowder was investigated and the sintering temperature that can make the full dense PLT specimen decreased to below 1050°C by using Bi_2O_3 powder as sintering agent. The pure PLT phase of perovskite structure was formed after MCP was conducted for 4 h and the average size of the particles was approximately 20 nm. After sintered at 1050 and 1150°C, the relative density of PLT was about 93.84 and 95.78%, respectively. The density of PLT increased with adding Bi_2O_3 and the specimen with the relative densitiy over 96% were fabricated below 1050°C when 2 wt% of Bi_2O_3 was added.
TOP
 KPMI
KPMI


 First
First Prev
Prev


