Search
- Page Path
- HOME > Search
- [Korean]
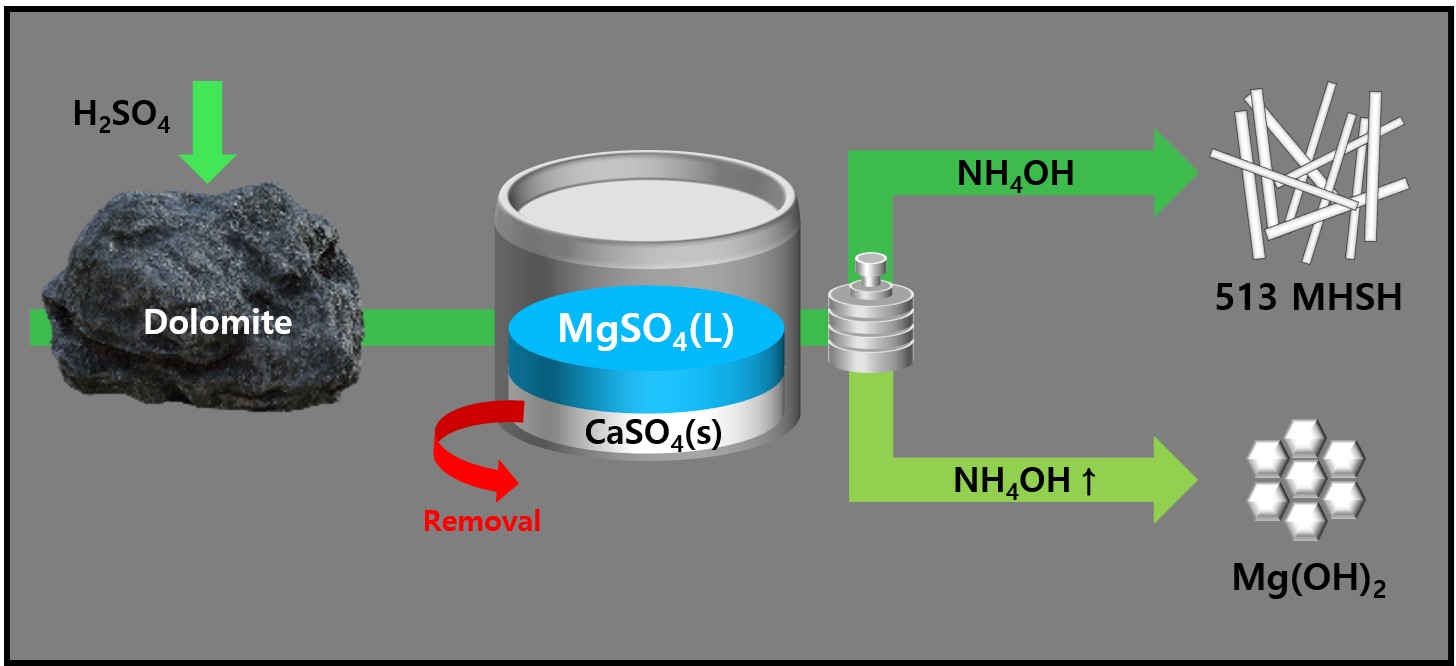
- Synthesis and Morphology Control of Needle Type 513 MHSH and Mg(OH)2 from Dolomite
- Jiyeon Kim, HyunSeung Shim, Seong-Ju Hwang, YooJin Kim
- J Powder Mater. 2025;32(5):399-405. Published online October 31, 2025
- DOI: https://doi.org/10.4150/jpm.2025.00227
- 559 View
- 8 Download
-
 Abstract
Abstract
 PDF
PDF - 513 magnesium hydroxide sulfate hydrate (MHSH) and Mg(OH)₂ were synthesized by controlling the pH and concentration using a domestic resource, dolomite (CaMg(CO3)2), as the raw material. The MgSO₄ was extracted by treating dolomite with sulfuric acid under various conditions. Hexagonal plate-shaped Mg(OH)₂ and needle-like 513 MHSH were synthesized under the hydrothermal condition. The morphology of the synthesized materials was controlled by adjusting the pH (SO42-/OH- ratio) and hydrothermal reaction time. As the pH of the solution increased, the formation of plate-like structures became dominant, whereas lower pH values (higher SO42- concentration) led to needle-like forms. The results of the 513 MHSH, which was synthesized using reagents and sea bittern, are consistent with the synthesis conditions, and we observed changes in the length and aspect ratio of the needle-shaped structure in response to adjusting the hydrothermal reaction time.
- [Korean]
- Inorganic Compound and Cycloserine Composite Particles for Improved Stability
- Dongwon Kim, Heeseo Kim, Hongjun Yoon, Hyuk Jun Cho, Sung Giu Jin
- J Powder Mater. 2024;31(2):126-131. Published online April 30, 2024
- DOI: https://doi.org/10.4150/jpm.2024.00002
- 6,728 View
- 32 Download
-
 Abstract
Abstract
 PDF
PDF - The aim of this study was to improve the chemical stability of cycloserine containing organic and inorganic compounds. Composite particles were manufactured with a 1:1 weight ratio of organic/inorganic compounds and cycloserine. The influence of organic/inorganic compounds on the stability of cycloserine was investigated under accelerated stress conditions at 60°C/75% RH for 24 hours. In addition, the properties of the composite particles were evaluated using differential scanning calorimetry (DSC), scanning electron microscopy (SEM), and the dissolution of the drug was assessed by preparing it as a hard capsule. Among the organic and inorganic compounds investigated, calcium hydroxide most improved the stability of cycloserine under accelerated stress conditions (53.3 ± 2.2% vs 1.7 ± 0.2%). DSC results confirmed the compatibility between calcium hydroxide and the cycloserine, and SEM results confirmed that it was evenly distributed around the cycloserine. Calcium hydroxide also showed more than 90% cycloserine dissolution within 15 minutes. Therefore, the calcium hydroxide and cycloserine composite particles may be candidates for cycloserine oral pharmaceuticals with enhanced drug stability.
- [Korean]
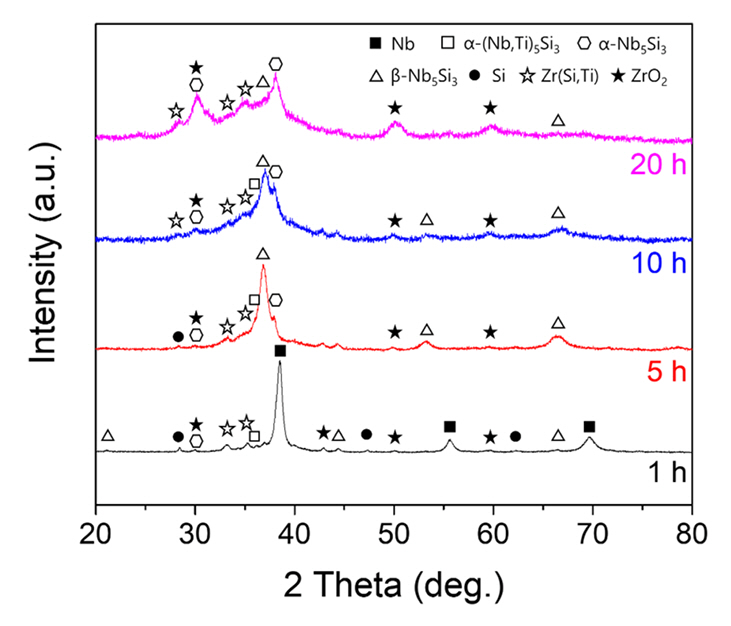
- Development of High-strength, High-temperature Nb-Si-Ti Alloys through Mechanical Alloying
- Jung-Joon Kim, Sang-Min Yoon, Deok-Hyun Han, Jongmin Byun, Young-Kyun Kim
- J Powder Mater. 2024;31(1):30-36. Published online February 28, 2024
- DOI: https://doi.org/10.4150/KPMI.2024.31.1.30
- 2,346 View
- 57 Download
- 1 Citations
-
 PDF
PDF -
Citations
Citations to this article as recorded by- Review of “Integrated Computer-Aided Process Engineering Session in the 17th International Symposium on Novel and Nano Materials (ISNNM, 14–18 November 2022)”
Yeon-Joo Lee, Pil-Ryung Cha, Hyoung-Seop Kim, Hyun-Joo Choi
MATERIALS TRANSACTIONS.2025; 66(1): 144. CrossRef
- Review of “Integrated Computer-Aided Process Engineering Session in the 17th International Symposium on Novel and Nano Materials (ISNNM, 14–18 November 2022)”
- [Korean]
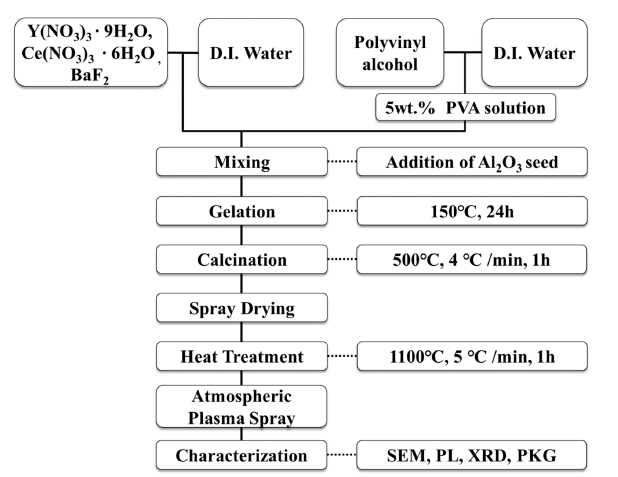
- Optical Properties of Spherical YAG:Ce3+ Phosphor Powders Synthesized by Atmospheric Plasma Spraying Method Appling PVA Solution Route and Domestic Aluminium Oxide Seed
- Yong-Hyeon Kim, Sang-Jin Lee
- J Powder Mater. 2023;30(5):424-430. Published online October 1, 2023
- DOI: https://doi.org/10.4150/KPMI.2023.30.5.424
- 1,121 View
- 4 Download
-
 Abstract
Abstract
 PDF
PDF YAG phosphor powders were fabricated by the atmospheric plasma spraying method with the spray-dried spherical YAG precursor. The YAG precursor slurry for the spray drying process was prepared by the PVA solution chemical processing utilizing a domestic easy-sintered aluminum oxide (Al2O3) powder as a seed. The homogenous and viscous slurry resulted in dense granules, not hollow or porous particles. The synthesized phosphor powders demonstrated a stable YAG phase, and excellent fluorescence properties of approximately 115% compared with commercial YAG:Ce3+ powder. The microstructure of the phosphor powder had a perfect spherical shape and an average particle s ize of a pprox imately 30 μm. As a r esult of t he PKG t est of t he YAG p hosphor p owder, t he s ynthesized phosphor powders exhibited an outstanding luminous intensity, and a peak wavelength was observed at 531 nm.
- [Korean]
- Fabrication and Evaluation of Levosulpiride-loaded Amorphous Spray-dried Microparticle for Improved Solubility
- Sung Giu Jin
- J Powder Mater. 2023;30(1):47-52. Published online February 1, 2023
- DOI: https://doi.org/10.4150/KPMI.2023.30.1.47
- 926 View
- 6 Download
-
 Abstract
Abstract
 PDF
PDF The purpose of this study is to develop and evaluate amorphous spray-dried microparticles (SDM) containing levosulpiride to increase its solubility. SDM are prepared via solvent evaporation using polyvinylpyrrolidone (PVP) as the water-soluble polymer and Cremophor RH40 as the surfactant. The SDM is prepared by varying the amounts of PVP and Cremophor RH40, and its physicochemical properties, solubility, and dissolution are confirmed. All levosulpiride-loaded SDMs converted the crystalline drug into an amorphous form, significantly improving drug solubility and dissolution compared with the drug alone. SDM consisting of drug/PVP/Cremophor RH40 in a weight ratio of 5:10:3, with increased solubility (720 ± 36 vs. 1822 ± 51 μg/mL) and dissolution rate (10.3 ± 2.2 vs. 92.6 ± 6.0%) compared with drug alone, shows potential as a commercial drug for improved oral bioavailability of levosulpiride.
- [Korean]
- Synthesis and Optical Property of (GaN)1-x(ZnO)x Nanoparticles Using an Ultrasonic Spray Pyrolysis Process and Subsequent Chemical Transformation
- Jeong Hyun Kim, Cheol-Hui Ryu, Myungjun Ji, Yomin Choi, Young-In Lee
- J Korean Powder Metall Inst. 2021;28(2):143-149. Published online April 1, 2021
- DOI: https://doi.org/10.4150/KPMI.2021.28.2.143
- 865 View
- 2 Download
-
 Abstract
Abstract
 PDF
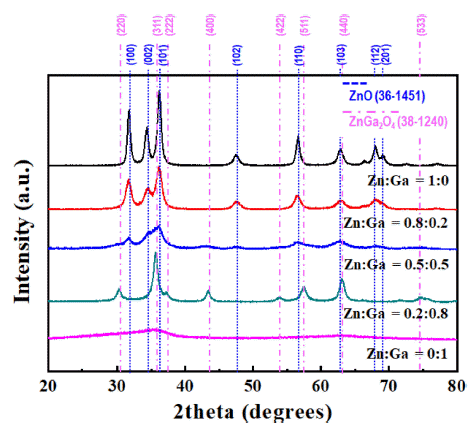
PDF In this study, (GaN)1-x(ZnO)x solid solution nanoparticles with a high zinc content are prepared by ultrasonic spray pyrolysis and subsequent nitridation. The structure and morphology of the samples are investigated by X-ray diffraction (XRD), field-emission scanning electron microscopy, and energy-dispersive X-ray spectroscopy. The characterization results show a phase transition from the Zn and Ga-based oxides (ZnO or ZnGa2O4) to a (GaN)1-x (ZnO)x solid solution under an NH3 atmosphere. The effect of the precursor solution concentration and nitridation temperature on the final products are systematically investigated to obtain (GaN)1-x(ZnO)x nanoparticles with a high Zn concentration. It is confirmed that the powder synthesized from the solution in which the ratio of Zn and Ga was set to 0.8:0.2, as the initial precursor composition was composed of about 0.8-mole fraction of Zn, similar to the initially set one, through nitriding treatment at 700°C. Besides, the synthesized nanoparticles exhibited the typical XRD pattern of (GaN)1-x(ZnO)x, and a strong absorption of visible light with a bandgap energy of approximately 2.78 eV, confirming their potential use as a hydrogen production photocatalyst.
- [Korean]
- Analysis of Wafer Cleaning Solution Characteristics and Metal Dissolution Behavior according to the Addition of Chelating Agent
- Myungsuk Kim, Keunhyuk Ryu, Kun-Jae Lee
- J Korean Powder Metall Inst. 2021;28(1):25-30. Published online February 1, 2021
- DOI: https://doi.org/10.4150/KPMI.2021.28.1.25

- 1,144 View
- 19 Download
-
 Abstract
Abstract
 PDF
PDF The surface of silicon dummy wafers is contaminated with metallic impurities owing to the reaction with and adhesion of chemicals during the oxidation process. These metallic impurities negatively affect the device performance, reliability, and yield. To solve this problem, a wafer-cleaning process that removes metallic impurities is essential. RCA (Radio Corporation of America) cleaning is commonly used, but there are problems such as increased surface roughness and formation of metal hydroxides. Herein, we attempt to use a chelating agent (EDTA) to reduce the surface roughness, improve the stability of cleaning solutions, and prevent the re-adsorption of impurities. The bonding between the cleaning solution and metal powder is analyzed by referring to the Pourbaix diagram. The changes in the ionic conductivity, H2O2 decomposition behavior, and degree of dissolution are checked with a conductivity meter, and the changes in the absorbance and particle size before and after the reaction are confirmed by ultraviolet-visible spectroscopy (UV-vis) and dynamic light scattering (DLS) analyses. Thus, the addition of a chelating agent prevents the decomposition of H2O2 and improves the life of the silicon wafer cleaning solution, allowing it to react smoothly with metallic impurities.
- [Korean]
- Synthesis and Nucleation Behavior of MoO3 Nano Particles with Concentration of Precursors
- Seyoung Lee, Namhun Kwon, Jaeseok Roh, Kun-Jae Lee
- J Korean Powder Metall Inst. 2020;27(5):394-400. Published online October 1, 2020
- DOI: https://doi.org/10.4150/KPMI.2020.27.5.394

- 1,800 View
- 9 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF Molybdenum trioxide (MoO3) is used in various applications including sensors, photocatalysts, and batteries owing to its excellent ionic conductivity and thermal properties. It can also be used as a precursor in the hydrogen reduction process to obtain molybdenum metals. Control of the parameters governing the MoO3 synthesis process is extremely important because the size and shape of MoO3 in the reduction process affect the shape, size, and crystallization of Mo metal. In this study, we fabricated MoO3 nanoparticles using a solution combustion synthesis (SCS) method that utilizes an organic additive, thereby controlling their morphology. The nucleation behavior and particle morphology were confirmed using ultraviolet-visible spectroscopy (UV-vis) and field emission scanning electron microscopy (FE-SEM). The concentration of the precursor (ammonium heptamolybdate tetrahydrate) was adjusted to be 0.1, 0.2, and 0.4 M. Depending on this concentration, different nucleation rates were obtained, thereby resulting in different particle morphologies.
-
Citations
Citations to this article as recorded by- Characterization of Compacted and Pressureless Sintered Parts for Molybdenum Oxide Powder according to Hydrogen Reduction Temperature
Jong Hoon Lee, Kun-Jae Lee
Journal of Powder Materials.2024; 31(4): 336. CrossRef
- Characterization of Compacted and Pressureless Sintered Parts for Molybdenum Oxide Powder according to Hydrogen Reduction Temperature
- [Korean]
- Recent Development in Fabrication and Control of Layered-Double Hydroxide Nanostructures
- Chan-Woo Jeon, Il-Kyu Park
- J Korean Powder Metall Inst. 2018;25(6):514-522. Published online December 1, 2018
- DOI: https://doi.org/10.4150/KPMI.2018.25.6.514
- 2,127 View
- 31 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
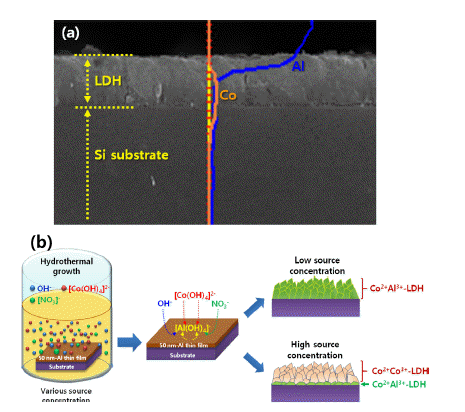
PDF Layered-double hydroxide (LDH)-based nanostructures offer the two-fold advantage of being active catalysts with incredibly large specific surface areas. As such, they have been studied extensively over the last decade and applied in roles as diverse as light source, catalyst, energy storage mechanism, absorber, and anion exchanger. They exhibit a unique lamellar structure consisting of a wide variety of combinations of metal cations and various anions, which determine their physical and chemical performances, and make them a popular research topic. Many reviewed papers deal with these unique properties, synthetic methods, and applications. Most of them, however, are focused on the form-factor of nanopowder, as well as on the control of morphologies via one-step synthetic methods. LDH nanostructures need to be easy to control and fabricate on rigid substrates such as metals, semiconductors, oxides, and insulators, to facilitate more viable applications of these nanostructures to various solid-state devices. In this review, we explore ways to grow and control the various LDH nanostructures on rigid substrates.
-
Citations
Citations to this article as recorded by- Review of Domestic Research Trends on Layered Double Hydroxide (LDH) Materials: Based on Research Articles in Korean Citation Index (KCI)
Seon Yong Lee, YoungJae Kim, Young Jae Lee
Economic and Environmental Geology.2023; 56(1): 23. CrossRef
- Review of Domestic Research Trends on Layered Double Hydroxide (LDH) Materials: Based on Research Articles in Korean Citation Index (KCI)
- [Korean]
- Research Trends in Powder Materials for Solution-based Transparent Conducting Electrode
- Bon-Ryul Koo, Hyo-Jin Ahn
- J Korean Powder Metall Inst. 2017;24(2):153-163. Published online April 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.2.153
- 901 View
- 4 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
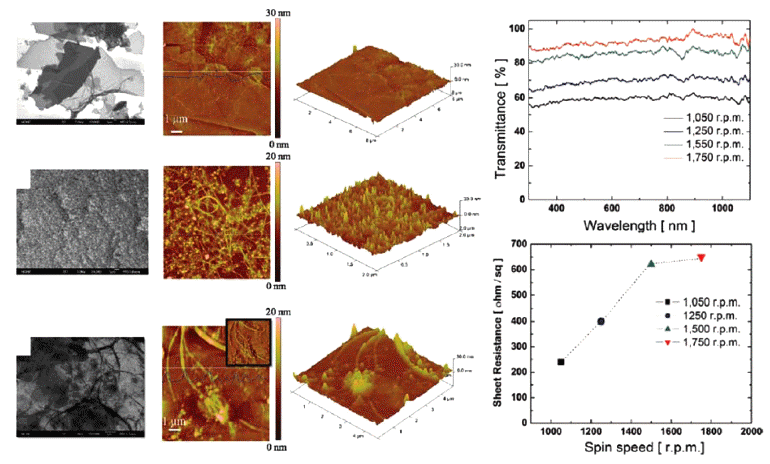
PDF Transparent conducting electrodes (TCEs) are attracting considerable attention as an important component for emerging optoelectronic applications such as liquid crystal displays, touch panels, and solar cells owing to their attractive combination of low resistivity (< 10-3 Ω cm) and high transparency (>80%) in the visible region. The solutionbased process has unique properties of an easy fabrication procedure, scalability, and low cost compared to the conventional vacuum-based process and may prove to be a useful process for fabricating TCEs for future optoelectronic applications demanding large scale and flexibility. In this paper, we focus on the introduction of a solution-based process for TCEs. In addition, we consider the powder materials used to fabricate solution-based TCEs and strategies to improve their transparent conducting properties.
-
Citations
Citations to this article as recorded by- Electrically conductive and anti-corrosive coating on copper foil assisted by polymer-nanocomposites embedded with graphene
Han Kim, Hyemin Lee, Hyo-Ryoung Lim, Hong-Baek Cho, Yong-Ho Choa
Applied Surface Science.2019; 476: 123. CrossRef
- Electrically conductive and anti-corrosive coating on copper foil assisted by polymer-nanocomposites embedded with graphene
- [Korean]
- Preparation of Ni(OH)2 Hollow Spheres by Solvent Displacement Crystallization Using Micro-Injection Device
- Seiki Kim, Kyungsoo Park, Kwang-Il Jung
- J Korean Powder Metall Inst. 2016;23(4):311-316. Published online August 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.4.311
- 1,302 View
- 9 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
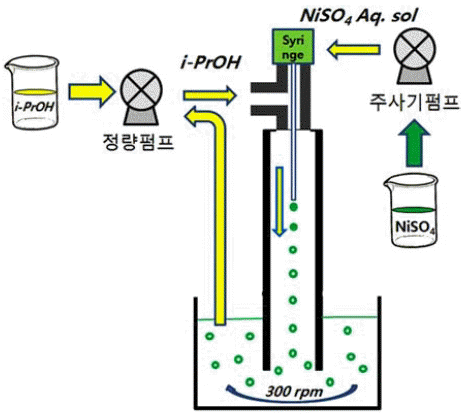
PDF Ni(OH)2 hollow spheres have been prepared by solvent displacement crystallization using a micro-injection device, and the effect of process parameters such as concentration and the relative ratio of the injection speed of the precursor solution, which is an aqueous solution of NiSO4·6H2O, to isopropyl alcohol of displacement solvent have been investigated. The crystal phases after NaOH treatment are in the β-phase for all process parameters. A higher concentration of NiSO4·6H2O aqueous solution is injected by a micro-injection device and bigger Ni(OH)2 hollow spheres with a narrower particle size distribution are formed. The crystallinity and hardness of the as-obtained powder are so poor that hydrothermal treatment of the as-obtained Ni(OH)2 at 120°C for 24 h in distilled water is performed in order to greatly improve the crystallinity. It is thought that a relative ratio of the injection speed of NiSO4·6H2O to that of isopropyl alcohol of at least more than 1 is preferable to synthesize Ni(OH)2 hollow spheres. It is confirmed that this solution- based process is very effective in synthesizing ceramic hollow spheres by simple adjustment of the process parameters such as the concentration and the injection speed.
-
Citations
Citations to this article as recorded by- The Effects of Hexamethylenetetramine Concentration on the Structural and Electrochemical Performances of Ni(OH)2 Powder for Pseudocapacitor Applications
Dong Yeon Kim, Young-Min Jeong, Seong-Ho Baek, Injoon Son
Journal of Korean Powder Metallurgy Institute.2019; 26(3): 231. CrossRef
- The Effects of Hexamethylenetetramine Concentration on the Structural and Electrochemical Performances of Ni(OH)2 Powder for Pseudocapacitor Applications
- [Korean]
- Analyses of Creep Properties of Ni-base Superalloy Powders as Cooling Rate after Solid Solution Heat Treatment
- Chan Jun, Youngseon Lee, Byeong Beom Bae, Hong-Kyu Kim, Seong Suk Hong, Donghoon Kim, Jondo Yun, Eun Yoo Yoon
- J Korean Powder Metall Inst. 2016;23(3):247-253. Published online June 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.3.247
- 1,073 View
- 2 Download
-
 Abstract
Abstract
 PDF
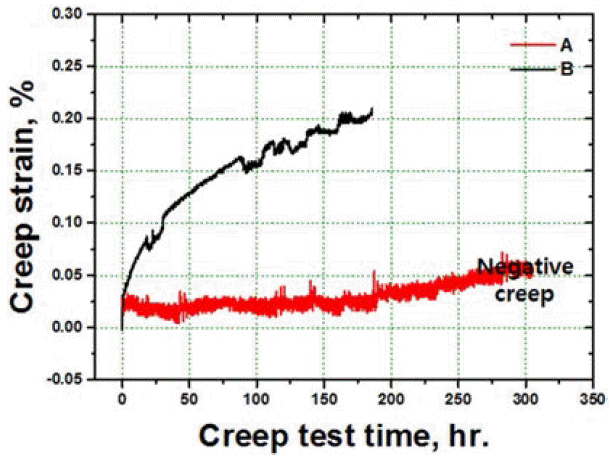
PDF In this study, solid solution heat treatment of consolidated nickel-based superalloy powders is carried out by hot isotactic pressing. The effects of the cooling rate of salt quenching, and air cooling on the microstructures and the mechanical properties of the specimens are analyzed . The specimen that is air cooled shows the formation of serrated grain boundaries due to their obstruction by the carbide particles. Moreover, the specimen that is salt quenched shows higher strength than the one that is air cooled due to the presence of fine and close-packed tertiary gamma prime phase. The tensile elongation at high temperatures improves due to the presence of grain boundary serrations in the specimen that is air cooled. On the contrary, the specimen that is salt quenched and consists of unserrated grain boundaries shows better creep properties than the air cooled specimen with the serrated grain boundaries, due to the negative creep phenomenon.
- [English]
- Production of Porous Metallic Glass Granule by Optimizing Chemical Processing
- Song-Yi Kim, Bo-Kyung Guem, Min-Ha Lee, Taek-Soo Kim, Jurgen Eckert, Bum-Sung Kim
- J Korean Powder Metall Inst. 2014;21(4):251-255. Published online August 1, 2014
- DOI: https://doi.org/10.4150/KPMI.2014.21.4.251
- 1,356 View
- 2 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
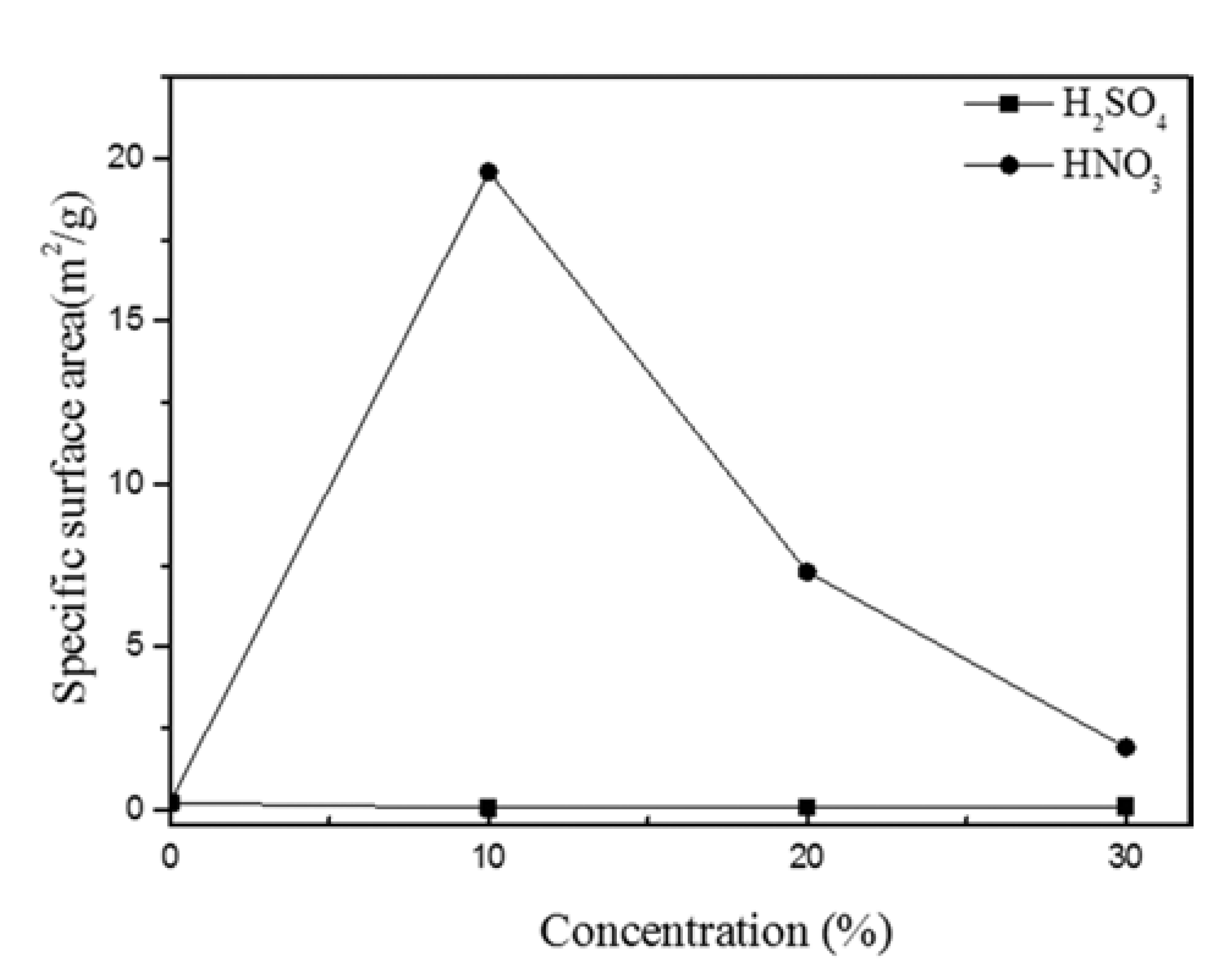
PDF In this study, we optimized dissolution the dissolution conditions of porous amorphous powder to have high specific surface area. Porous metallic glass(MG) granules were fabricated by selective phase dissolution, in which brass is removed from a composite powder consisting of MG and 40 vol.% brass. Dissolution was achieved through various concentrations of H2SO4 and HNO3, with HNO3 proving to have the faster reaction kinetics. Porous powders were analyzed by differential scanning calorimetry to observe crystallization behavior. The Microstructure of milled powder and dissolved powder was analyzed by scanning electron microscope. To check for residual in the dissolved powder after dissolution, energy dispersive X-ray spectroscory and elemental mapping was conducted. It was confirmed that the MG/brass composite powder dissolved in 10% HNO3 produced a porous MG granule with a relatively high specific surface area of 19.60 m2/g. This proved to be the optimum dissolution condition in which both a porous internal granule structure and amorphous phase were maintained. Consequently, porous MG granules were effectively fabricated and applications of such structures can be expanded.
-
Citations
Citations to this article as recorded by- Enhanced wear resistivity of a Zr-based bulk metallic glass processed by high-pressure torsion under reciprocating dry conditions
Soo-Hyun Joo, Dong-Hai Pi, Jing Guo, Hidemi Kato, Sunghak Lee, Hyoung Seop Kim
Metals and Materials International.2016; 22(3): 383. CrossRef
- Enhanced wear resistivity of a Zr-based bulk metallic glass processed by high-pressure torsion under reciprocating dry conditions
TOP
 KPMI
KPMI


 First
First Prev
Prev


