Search
- Page Path
- HOME > Search
- [Korean]
- Effect of Powder Preparation Method on the Microstructural Characteristics of Sintered W-7Ni-3Cu Heavy Alloy
- Youngmin Kim, Ji Young Kim, Minju Son, Wonyong Kwon, Eui Seon Lee, Sung-Tag Oh
- J Powder Mater. 2026;33(2):113-118. Published online April 30, 2026
- DOI: https://doi.org/10.4150/jpm.2026.00038

- 765 View
- 8 Download
-
 Abstract
Abstract
 PDF
PDF - The effect of powder characteristics and sintering temperature on the properties of W-7Ni-3Cu is investigated. The heavy alloy powders were prepared by ball milling and hydrogen reduction of elemental metal or metal oxide powders. Microstructural analysis revealed that the powder mixtures reduced by hydrogen at 800oC consist of a trace amount of Ni4W phase along with the metal W phase and Ni-Cu solid solution. Additionally, compared to metal powder, the powder mixture using oxide as raw material exhibited a relatively fine particle size. The W-7Ni-3Cu alloys sintered using oxide powders had relative density of over 99%, whereas the specimens using metal powders as a raw material showed relatively low values of 87.8~98.2%. The Vickers hardness of the sintered specimens using oxide powder was 3.34–3.92 GPa, which was higher than that of 2.39–3.22 GPa measured when using metal powders. The observed results can be attributed to the relatively high density and the reduced grain size.
- [Korean]
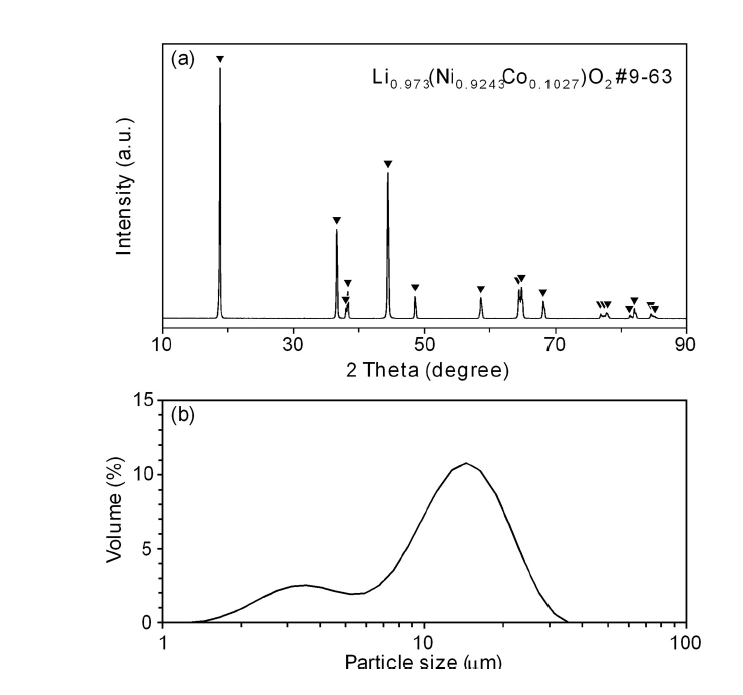
- Hydrogen Reduction Behavior of NCM-based Lithium-ion Battery Cathode Materials
- So-Yeong Lee, So-Yeon Lee, Dae-Hyeon Lee, Ho-Sang Sohn
- J Powder Mater. 2024;31(2):163-168. Published online April 30, 2024
- DOI: https://doi.org/10.4150/jpm.2024.00017
- 2,073 View
- 51 Download
- 2 Citations
-
 Abstract
Abstract
 PDF
PDF - As the demand for lithium-ion batteries for electric vehicles is increasing, it is important to recover valuable metals from waste lithium-ion batteries. In this study, the effects of gas flow rate and hydrogen partial pressure on hydrogen reduction of NCM-based lithium-ion battery cathode materials were investigated. As the gas flow rate and hydrogen partial pressure increased, the weight loss rate increased significantly from the beginning of the reaction due to the reduction of NiO and CoO by hydrogen. At 700 °C and hydrogen partial pressure above 0.5 atm, Ni and Li2O were produced by hydrogen reduction. From the reduction product and Li recovery rate, the hydrogen reduction of NCM-based cathode materials was significantly affected by hydrogen partial pressure. The Li compounds recovered from the solution after water leaching of the reduction products were LiOH, LiOH·H2O, and Li2CO3, with about 0.02 wt% Al as an impurity.
-
Citations
Citations to this article as recorded by- Research Progress on Prior Lithium Extraction from Spent Lithium-Ion Battery Cathode Materials via Pyrometallurgical Roasting
Zhanyong Guo, Xiangrui Ren, Zihan Zhang, Zhen Feng, Fachuang Li
Sustainability.2026; 18(8): 4026. CrossRef - Reduction Roasting of Black Mass Recovered from NCM-based Spent Lithium-ion Batteries Using CH4 Gas
Sang-Yeop Lee, Jae-Ho Hwang, Ho-Sang Sohn
Resources Recycling.2025; 34(5): 93. CrossRef
- Research Progress on Prior Lithium Extraction from Spent Lithium-Ion Battery Cathode Materials via Pyrometallurgical Roasting
- [Korean]
- Fabrication of Bi2Te2.5Se0.5 by Combining Oxide-reduction and Compressive-forming Process and Its Thermoelectric Properties
- Young Soo Lim, Gil-Geun Lee
- J Powder Mater. 2024;31(1):50-56. Published online February 28, 2024
- DOI: https://doi.org/10.4150/KPMI.2024.31.1.50
- 1,334 View
- 22 Download
- [Korean]
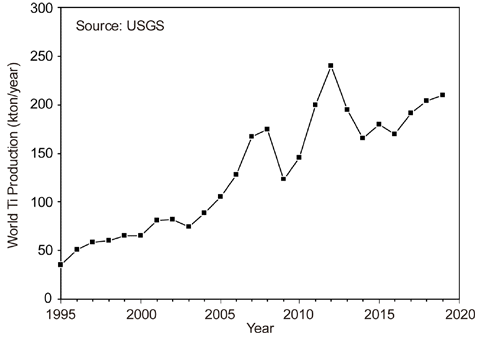
- Current Status of Titanium Smelting Technology for Powder Metallurgy
- Ho-Sang Sohn
- J Korean Powder Metall Inst. 2021;28(2):164-172. Published online April 1, 2021
- DOI: https://doi.org/10.4150/KPMI.2021.28.2.164
- 1,278 View
- 9 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF Titanium is the ninth most abundant element in the Earth’s crust and is the fourth most abundant structural metal after aluminum, iron, and magnesium. It exhibits a higher specific strength than steel along with an excellent corrosion resistance, highlighting the promising potential of titanium as a structural metal. However, titanium is difficult to extract from its ore and is classified as a rare metal, despite its abundance. Therefore, the production of titanium is exceedingly low compared to that of common metals. Titanium is conventionally produced as a sponge by the Kroll process. For powder metallurgy (PM), hydrogenation-dehydrogenation (HDH) of the titanium sponge or gas atomization of the titanium bulk is required. Therefore, numerous studies have been conducted on smelting, which replaces the Kroll process and produces powder that can be used directly for PM. In this review, the Kroll process and new smelting technologies of titanium for PM, such as metallothermic, electrolytic, and hydrogen reduction of TiCl4 and TiO2 are discussed.
-
Citations
Citations to this article as recorded by- Enhancing corrosion resistance of Ti-based amorphous alloy powders via misch metal addition
Yeon Joo Lee, Hyokyung Sung, Jae Bok Seol, Kisub Cho, Hwi Jun Kim, Hyunjoo Choi
Powder Metallurgy.2025; 68(3): 230. CrossRef
- Enhancing corrosion resistance of Ti-based amorphous alloy powders via misch metal addition
- [Korean]
- Synthesis of Porous Cu-Co using Freeze Drying Process of Camphene Slurry with Oxide Composite Powders
- Gyuhwi Lee, Ju-Yeon Han, Sung-Tag Oh
- J Korean Powder Metall Inst. 2020;27(3):193-197. Published online June 1, 2020
- DOI: https://doi.org/10.4150/KPMI.2020.27.3.193

- 936 View
- 3 Download
-
 Abstract
Abstract
 PDF
PDF Porous Cu-14 wt% Co with aligned pores is produced by a freeze drying and sintering process. Unidirectional freezing of camphene slurry with CuO-Co3O4 powders is conducted, and pores in the frozen specimens are generated by sublimation of the camphene crystals. The dried bodies are hydrogen-reduced at 500°C and sintered at 800°C for 1 h. The reduction behavior of the CuO-Co3O4 powder mixture is analyzed using a temperature-programmed reduction method in an Ar-10% H2 atmosphere. The sintered bodies show large and aligned parallel pores in the camphene growth direction. In addition, small pores are distributed around the internal walls of the large pores. The size and fraction of the pores decrease as the amount of solid powder added to the slurry increases. The change in pore characteristics according to the amount of the mixed powder is interpreted to be due to the rearrangement and accumulation behavior of the solid particles in the freezing process of the slurry.
- [Korean]
- Hydrogen Reduction Behavior and Microstructure Characteristics of Ball-milled CuO-Co3O4 Powder Mixtures
- Ju-Yeon Han, Gyuhwi Lee, Hyunji Kang, Sung-Tag Oh
- J Korean Powder Metall Inst. 2019;26(5):410-414. Published online October 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.5.410

- 977 View
- 3 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF The hydrogen reduction behavior of the CuO-Co3O4 powder mixture for the synthesis of the homogeneous Cu-15at%Co composite powder has been investigated. The composite powder is prepared by ball milling the oxide powders, followed by a hydrogen reduction process. The reduction behavior of the ball-milled powder mixture is analyzed by X-ray diffraction (XRD) and temperature-programmed reduction at different heating rates in an Ar-10%H2 atmosphere. The scanning electron microscopy and XRD results reveal that the hydrogen-reduced powder mixture is composed of fine agglomerates of nanosized Cu and Co particles. The hydrogen reduction kinetics is studied by determining the degree of peak shift as a function of the heating rate. The activation energies for the reduction of the oxide powders estimated from the slopes of the Kissinger plots are 58.1 kJ/mol and 65.8 kJ/mol, depending on the reduction reaction: CuO to Cu and Co3O4 to Co, respectively. The measured temperature and activation energy for the reduction of Co3O4 are explained on the basis of the effect of pre-reduced Cu particles.
-
Citations
Citations to this article as recorded by- Synthesis of Porous Cu-Co using Freeze Drying Process of Camphene Slurry with Oxide Composite Powders
Gyuhwi Lee, Ju-Yeon Han, Sung-Tag Oh
Journal of Korean Powder Metallurgy Institute.2020; 27(3): 193. CrossRef
- Synthesis of Porous Cu-Co using Freeze Drying Process of Camphene Slurry with Oxide Composite Powders
- [English]
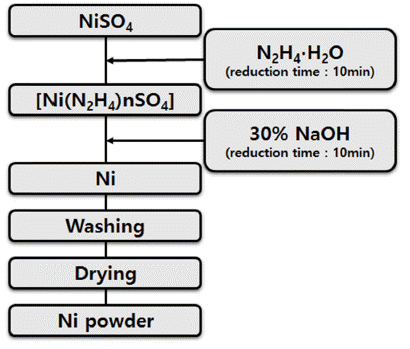
- Synthesis of Nanosized Nickel Particle from Spent Cathodic Material Containing Lithium
- Jei-Pil Wang
- J Korean Powder Metall Inst. 2019;26(4):340-344. Published online August 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.4.340
- 1,280 View
- 5 Download
-
 Abstract
Abstract
 PDF
PDF Due to the rapid development of electricity, electronics, information communication, and biotechnology in recent years, studies are actively being conducted on nanopowders as it is required not only for high strengthening but also for high-function powder with electric, magnetic, and optical properties. Nonetheless, studies on nickel nanopowders are rare. In this study of the synthesis of nickel nanoparticles from LiNiO2 (LNO), which is a cathode active material, we have synthesized the nanosized nickel powder by the liquid reduction process of NiSO4 obtained through the leaching and purification of LNO. Moreover, we have studied the reduction reaction rate according to the temperature change of liquid phase reduction and the change of particle size as a function of NaOH addition amount using hydrazine monohydrate (N2H4·H2O) and NaOH.
- [Korean]
- Study on the Optimization of Reduction Conditions for Samarium-Cobalt Nanofiber Preparation
- Jimin Lee, Jongryoul Kim, Yong-Ho Choa
- J Korean Powder Metall Inst. 2019;26(4):334-339. Published online August 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.4.334

- 701 View
- 2 Download
-
 Abstract
Abstract
 PDF
PDF To meet the current demand in the fields of permanent magnets for achieving a high energy density, it is imperative to prepare nano-to-microscale rare-earth-based magnets with well-defined microstructures, controlled homogeneity, and magnetic characteristics via a bottom-up approach. Here, on the basis of a microstructural study and qualitative magnetic measurements, optimized reduction conditions for the preparation of nanostructured Sm-Co magnets are proposed, and the elucidation of the reduction-diffusion behavior in the binary phase system is clearly manifested. In addition, we have investigated the microstructural, crystallographic, and magnetic properties of the Sm-Co magnets prepared under different reduction conditions, that is, H2 gas, calcium, and calcium hydride. This work provides a potential approach to prepare high-quality Sm-Co-based nanofibers, and moreover, it can be extended to the experimental design of other magnetic alloys.
- [Korean]
- Study on the Optimization of Reduction Conditions for Samarium-Cobalt Nanofiber Preparation
- Jimin Lee, Jongryoul Kim, Yong-Ho Choa
- J Korean Powder Metall Inst. 2019;26(4):334-339. Published online August 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.4.334

- 629 View
- 2 Download
-
 Abstract
Abstract
 PDF
PDF To meet the current demand in the fields of permanent magnets for achieving a high energy density, it is imperative to prepare nano-to-microscale rare-earth-based magnets with well-defined microstructures, controlled homogeneity, and magnetic characteristics via a bottom-up approach. Here, on the basis of a microstructural study and qualitative magnetic measurements, optimized reduction conditions for the preparation of nanostructured Sm-Co magnets are proposed, and the elucidation of the reduction-diffusion behavior in the binary phase system is clearly manifested. In addition, we have investigated the microstructural, crystallographic, and magnetic properties of the Sm-Co magnets prepared under different reduction conditions, that is, H2 gas, calcium, and calcium hydride. This work provides a potential approach to prepare high-quality Sm-Co-based nanofibers, and moreover, it can be extended to the experimental design of other magnetic alloys.
- [Korean]
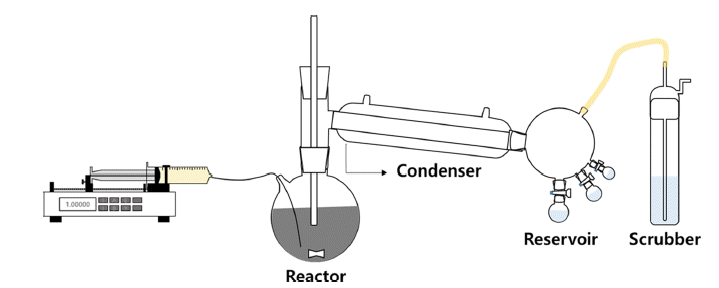
- Study on Manufacture of High Purity TiCl4 and Synthesis of High Purity Ti Powders
- Jieun Lee, Jin-Ho Yoon, Chan Gi Lee
- J Korean Powder Metall Inst. 2019;26(4):282-289. Published online August 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.4.282
- 1,750 View
- 33 Download
-
 Abstract
Abstract
 PDF
PDF Ti has received considerable attention for aerospace, vehicle, and semiconductor industry applications because of its acid-resistant nature, low density, and high mechanical strength. A common precursor used for preparing Ti materials is TiCl4. To prepare high-purity TiCl4, a process based on the removal of VOCl3 has been widely applied. However, VOCl3 removal by distillation and condensation is difficult because of the similar physical properties of TiCl4 and VOCl3. To circumvent this problem, in this study, we have developed a process for VOCl3 removal using Cu powder and mineral oil as purifying agents. The effects of reaction time and temperature, and ratio of purifying agents on the VOCl3 removal efficiency are investigated by chemical and structural measurements. Clear TiCl4 is obtained after the removal of VOCl3. Notably, complete removal of VOCl3 is achieved with 2.0 wt% of mineral oil. Moreover, the refined TiCl4 is used as a precursor for the synthesis of Ti powder. Ti powder is fabricated by a thermal reduction process at 1,100ºC using an H2-Ar gas mixture. The average size of the Ti powder particles is in the range of 1-3 μm.
- [Korean]
- Fabrication of Porous Ni by Freeze Drying and Hydrogen Reduction of NiO/Camphene Slurry
- Jae-Hun Jeong, Sung-Tag Oh, Chang-Yong Hyun
- J Korean Powder Metall Inst. 2019;26(1):6-10. Published online February 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.1.6

- 1,093 View
- 2 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF In this study, freeze drying of a porous Ni with unidirectionally aligned pore channels is accomplished by using a NiO powder and camphene. Camphene slurries with NiO content of 5 and 10 vol% are prepared by mixing them with a small amount of dispersant at 50°C. Freezing of a slurry is performed at -25°C while the growth direction of the camphene is unidirectionally controlled. Pores are generated subsequently by sublimation of the camphene during drying in air for 48 h. The green bodies are hydrogen-reduced at 400°C and then sintered at 800°C and 900°C for 1 h. X-ray diffraction analysis reveals that the NiO powder is completely converted to the Ni phase without any reaction phases. The sintered samples show large pores that align parallel pores in the camphene growth direction as well as small pores in the internal walls of large pores. The size of large and small pores decreases with increasing powder content from 5 to 10 vol%. The influence of powder content on the pore structure is explained by the degree of powder rearrangement in slurry and the accumulation behavior of powders in the interdendritic spaces of solidified camphene.
-
Citations
Citations to this article as recorded by- Synthesis of Porous Cu-Co using Freeze Drying Process of Camphene Slurry with Oxide Composite Powders
Gyuhwi Lee, Ju-Yeon Han, Sung-Tag Oh
Journal of Korean Powder Metallurgy Institute.2020; 27(3): 193. CrossRef
- Synthesis of Porous Cu-Co using Freeze Drying Process of Camphene Slurry with Oxide Composite Powders
- [Korean]
- Fabrication of Porous Mo-Cu by Freeze Drying and Hydrogen Reduction of Metal Oxide Powders
- Hyunji Kang, Ju-Yeon Han, Sung-Tag Oh
- J Korean Powder Metall Inst. 2019;26(1):1-5. Published online February 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.1.1

- 1,260 View
- 4 Download
- 2 Citations
-
 Abstract
Abstract
 PDF
PDF In this study, porous Mo-5 wt% Cu with unidirectionally aligned pores is prepared by freeze drying of camphene slurry with MoO3-CuO powders. Unidirectional freezing of camphene slurry with dispersion stability is conducted at -25°C, and pores in the frozen specimens are generated by sublimation of the camphene crystals. The green bodies are hydrogen-reduced at 750°C and sintered at 1000°C for 1 h. X-ray diffraction analysis reveals that MoO3-CuO composite powders are completely converted to a Mo-and-Cu phase without any reaction phases by hydrogen reduction. The sintered bodies with the Mo-Cu phase show large and aligned parallel pores to the camphene growth direction as well as small pores in the internal walls of large pores. The pore size and porosity decrease with increasing composite powder content from 5 to 10 vol%. The change of pore characteristics is explained by the degree of powder rearrangement in slurry and the accumulation behavior of powders in the interdendritic spaces of solidified camphene.
-
Citations
Citations to this article as recorded by- Characteristic Evaluation of WC Hard Materials According to Ni Content Variation by a Pulsed Current Activated Sintering Process
Hyun-Kuk Park
Korean Journal of Materials Research.2020; 30(12): 672. CrossRef - Effect of α-lath size on the mechanical properties of Ti–6Al–4V using core time hydrogen heat treatment
Gye-Hoon Cho, Jung-Min Oh, Hanjung Kwon, Jae-Won Lim
Materials Science and Technology.2020; 36(7): 858. CrossRef
- Characteristic Evaluation of WC Hard Materials According to Ni Content Variation by a Pulsed Current Activated Sintering Process
- [Korean]
- Fabrication of Metallic Tantalum Powder by Magnesium-gas Reduction of Tantalum Oxide
- Dong-Won Lee
- J Korean Powder Metall Inst. 2018;25(5):390-394. Published online October 1, 2018
- DOI: https://doi.org/10.4150/KPMI.2018.25.5.390

- 1,095 View
- 14 Download
- 2 Citations
-
 Abstract
Abstract
 PDF
PDF Metallic tantalum powder is manufactured by reducing tantalum oxide (Ta2O5) with magnesium gas at 1,073–1,223 K in a reactor under argon gas. The high thermodynamic stability of magnesium oxide makes the reduction reaction from tantalum oxide into tantalum powder possible. The microstructure after the reduction reaction has the form of a mixture of tantalum and magnesium oxide, and the latter could be entirely eliminated by dissolving in weak hydrochloric acid. The powder size in SEM microstructure for the tantalum powder increases after acid leaching in the range of 50–300 nm, and its internal crystallite sizes are observed to be 11.5 to 24.7 nm with increasing reduction temperatures. Moreover, the optimized reduction temperature is found to be 1,173 K as the minimum oxygen concentration is approximately 1.3 wt.%.
-
Citations
Citations to this article as recorded by- A review of tantalum resources and its production
Xue WEI, Long-gong XIA, Zhi-hong LIU, Le-ru ZHANG, Qi-hou LI
Transactions of Nonferrous Metals Society of China.2023; 33(10): 3132. CrossRef - Valuable metal recovery from waste tantalum capacitors via cryogenic crushing-alkaline calcination-leaching process
Longgong Xia, Xue Wei, Hongjun Wang, Fengchun Ye, Zhihong Liu
Journal of Materials Research and Technology.2022; 16: 1637. CrossRef
- A review of tantalum resources and its production
- [Korean]
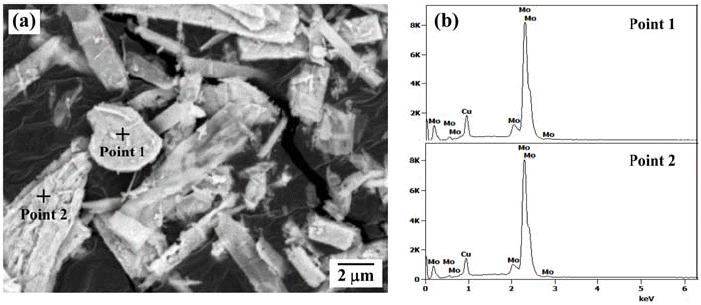
- Fabrication of Mo-Cu Powders by Ball Milling and Hydrogen Reduction of MoO3-CuO Powder Mixtures
- Hyunji Kang, Sung-Tag Oh
- J Korean Powder Metall Inst. 2018;25(4):322-326. Published online August 1, 2018
- DOI: https://doi.org/10.4150/KPMI.2018.25.4.322
- 1,206 View
- 3 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF The hydrogen reduction behavior of MoO3-CuO powder mixture for the synthesis of homogeneous Mo-20 wt% Cu composite powder is investigated. The reduction behavior of ball-milled powder mixture is analyzed by XRD and temperature programmed reduction method at various heating rates in Ar-10% H2 atmosphere. The XRD analysis of the heat-treated powder at 300°C shows Cu, MoO3, and Cu2MoO5 phases. In contrast, the powder mixture heated at 400°C is composed of Cu and MoO2 phases. The hydrogen reduction kinetic is evaluated by the amount of peak shift with heating rates. The activation energies for the reduction, estimated by the slope of the Kissinger plot, are measured as 112.2 kJ/mol and 65.2 kJ/mol, depending on the reduction steps from CuO to Cu and from MoO3 to MoO2, respectively. The measured activation energy for the reduction of MoO3 is explained by the effect of pre-reduced Cu particles. The powder mixture, hydrogen-reduced at 700°C, shows the dispersion of nano-sized Cu agglomerates on the surface of Mo powders.
-
Citations
Citations to this article as recorded by- Synthesis of Mo-Cu nanocomposite powder by hydrogen reduction of copper nitrate coated MoO3 powder mixture
Ji Won Choi, Ji Young Kim, Youngmin Kim, Eui Seon Lee, Sung-Tag Oh
Materials Letters.2024; 377: 137565. CrossRef - Fabrication of Porous Mo-Cu by Freeze Drying and Hydrogen Reduction of Metal Oxide Powders
Hyunji Kang, Ju-Yeon Han, Sung-Tag Oh
Journal of Korean Powder Metallurgy Institute.2019; 26(1): 1. CrossRef - Hydrogen Reduction Behavior and Microstructure Characteristics of Ball-milled CuO-Co3O4 Powder Mixtures
Ju-Yeon Han, Gyuhwi Lee, Hyunji Kang, Sung-Tag Oh
Journal of Korean Powder Metallurgy Institute.2019; 26(5): 410. CrossRef
- Synthesis of Mo-Cu nanocomposite powder by hydrogen reduction of copper nitrate coated MoO3 powder mixture
- [Korean]
- Fabrication of WC/Co composite powder from oxide of WC/Co hardmetal scrap by carbothermal reduction process
- Gil-Geun Lee, Young Soo Lim
- J Korean Powder Metall Inst. 2018;25(3):240-245. Published online June 1, 2018
- DOI: https://doi.org/10.4150/KPMI.2018.25.3.240

- 980 View
- 5 Download
-
 Abstract
Abstract
 PDF
PDF This study focuses on the fabrication of a WC/Co composite powder from the oxide of WC/Co hardmetal scrap using solid carbon in a hydrogen gas atmosphere for the recycling of WC/Co hardmetal. Mixed powders are manufactured by mechanically milling the oxide powder of WC-13 wt% Co hardmetal scrap and carbon black with varying powder/ball weight ratios. The oxide powder of WC-13 wt% Co hardmetal scrap consists of WO3 and CoWO4. The mixed powder mechanically milled at a lower powder/ball weight ratio (high mechanical milling energy) has a more rapid carbothermal reduction reaction in the formation of WC and Co phases compared with that mechanically milled at a higher powder/ball weight ratio (lower mechanical milling energy). The WC/Co composite powder is fabricated at 900°C for 6 h from the oxide of WC/Co hardmetal scrap using solid carbon in a hydrogen gas atmosphere. The fabricated WC/Co composite powder has a particle size of approximately 0.25-0.5 μm.
- [Korean]
- Study on the Recovery Silver and Nanoparticles Synthesis from LTCC By-products of Lowly Concentrated Silver
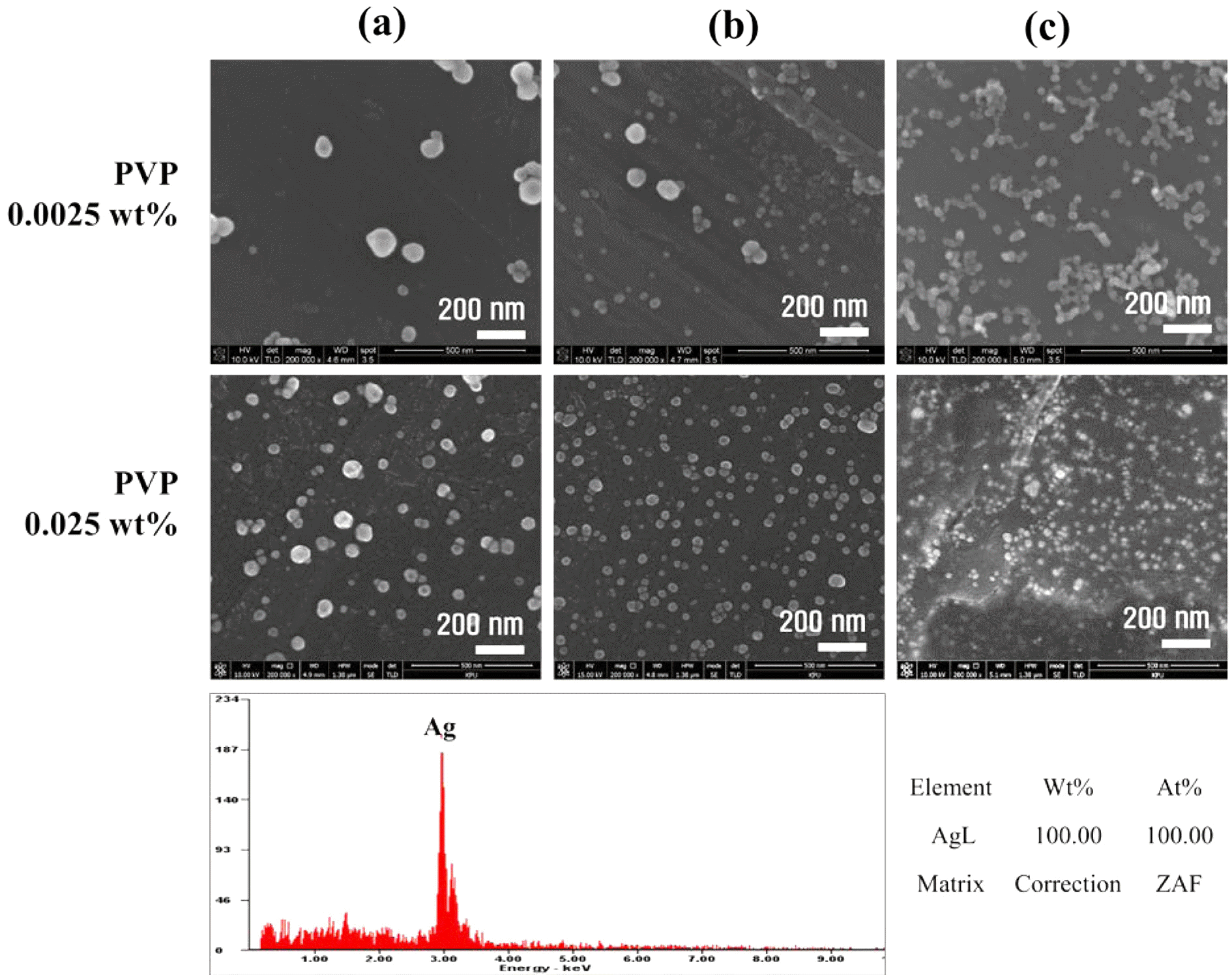
- Soyeong Joo, Nak-Kyoon Ahn, Chan Gi Lee, Jin-Ho Yoon
- J Korean Powder Metall Inst. 2018;25(3):232-239. Published online June 1, 2018
- DOI: https://doi.org/10.4150/KPMI.2018.25.3.232
- 1,056 View
- 5 Download
-
 Abstract
Abstract
 PDF
PDF In this paper, the recovery and nanoparticle synthesis of Ag from low temperature co-fired ceramic (LTCC) by-products are studied. The effect of reaction behavior on Ag leaching conditions from the LTCC by-products is confirmed. The optimum leaching conditions are determined to be: 5 M HNO3, a reaction temperature of 75°C, and a pulp density of 50 g/L at 60 min. For the selective recovery of Ag, the [Cl]/[Ag] equivalence ratio experiment is performed using added HCl; most of the Ag (more than 99%) is recovered. The XRD and MP-AES results confirm that the powder is AgCl and that impurities are at less than 1%. Ag nanoparticles are synthesized using a chemical reduction process for recycling, NaBH4 and PVP are used as reducing agents and dispersion stabilizers. UV-vis and FE-SEM results show that AgCl powder is precipitated and that Ag nanoparticles are synthesized. Ag nanoparticles of 100% Ag are obtained under the chemical reaction conditions.
- [Korean]
- Fabrication of Porous W-Ti by Freeze-Drying and Hydrogen Reduction of WO3-TiH2 Powder Mixtures
- Hyunji Kang, Sung Hyun Park, Sung-Tag Oh
- J Korean Powder Metall Inst. 2017;24(6):472-476. Published online December 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.6.472

- 774 View
- 1 Download
-
 Abstract
Abstract
 PDF
PDF Porous W-10 wt% Ti alloys are prepared by freeze-drying a WO3-TiH2/camphene slurry, using a sintering process. X-ray diffraction analysis of the heat-treated powder in an argon atmosphere shows the WO3 peak of the starting powder and reaction-phase peaks such as WO2.9, WO2, and TiO2 peaks. In contrast, a powder mixture heated in a hydrogen atmosphere is composed of the W and TiW phases. The formation of reaction phases that are dependent on the atmosphere is explained by a thermodynamic consideration of the reduction behavior of WO3 and the dehydrogenation reaction of TiH2. To fabricate a porous W-Ti alloy, the camphene slurry is frozen at -30°C, and pores are generated in the frozen specimens by the sublimation of camphene while drying in air. The green body is hydrogen-reduced and sintered at 1000°C for 1 h. The sintered sample prepared by freeze-drying the camphene slurry shows large and aligned parallel pores in the camphene growth direction, and small pores in the internal walls of the large pores. The strut between large pores consists of very fine particles with partial necking between them.
- [Korean]
- Effect of Powder Mixing Process on the Characteristics of Hybrid Structure Tungsten Powders with Nano-Micro Size
- Na-Yeon Kwon, Young-Keun Jeong, Sung-Tag Oh
- J Korean Powder Metall Inst. 2017;24(5):384-388. Published online October 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.5.384

- 1,152 View
- 3 Download
- 5 Citations
-
 Abstract
Abstract
 PDF
PDF The effect of the mixing method on the characteristics of hybrid-structure W powder with nano and micro sizes is investigated. Fine WO3 powders with sizes of ~0.6 μm, prepared by ball milling for 10 h, are mixed with pure W powder with sizes of 12 μm by various mixing process. In the case of simple mixing with ball-milled WO3 and micro sized W powders, WO3 particles are locally present in the form of agglomerates in the surface of large W powders, but in the case of ball milling, a relatively uniform distribution of WO3 particles is exhibited. The microstructural observation reveals that the ball milled WO3 powder, heat-treated at 750°C for 1 h in a hydrogen atmosphere, is fine W particles of ~200 nm or less. The powder mixture prepared by simple mixing and hydrogen reduction exhibits the formation of coarse W particles with agglomeration of the micro sized W powder on the surface. Conversely, in the powder mixture fabricated by ball milling and hydrogen reduction, a uniform distribution of fine W particles forming nano-micro sized hybrid structure is observed.
-
Citations
Citations to this article as recorded by- The Efficiency of Radiation Shielding Sheet to Reduce Radiation Exposure during C-arm Fluoroscopy
Hosang Jeon, Won Chul Shin, Hee Yun Seol, Yongkan Ki, Kyeong Baek Kim, Ki Seok Choo, Sang Don Lee, Suk-Woong Kang
Journal of the Korean Fracture Society.2023; 36(4): 111. CrossRef - Microstructure and Sintering Behavior of Fine Tungsten Powders Synthesized by Ultrasonic Spray Pyrolysis
Hyeonhui Jo, Jeong Hyun Kim, Young-In Lee, Young-Keun Jeong, Sung-Tag Oh
Korean Journal of Metals and Materials.2021; 59(5): 289. CrossRef - Facile phosphorus-embedding into SnS2 using a high-energy ball mill to improve the surface kinetics of P-SnS2 anodes for a Li-ion battery
Hongsuk Choi, Seungmin Lee, KwangSup Eom
Applied Surface Science.2019; 466: 578. CrossRef - Hydrogen reduction behavior and microstructural characteristics of WO3 and WO3-NiO powders
Hyunji Kang, Young-Keun Jeong, Sung-Tag Oh
International Journal of Refractory Metals and Hard Materials.2019; 80: 69. CrossRef - Fabrication of Densified W-Ti by Reaction Treatment and Spark Plasma Sintering of WO3-TiH2 Powder Mixtures
Hyunji Kang, Heun Joo Kim, Ju-Yeon Han, Yunju Lee, Young-Keun Jeong, Sung-Tag Oh
Korean Journal of Materials Research.2018; 28(9): 511. CrossRef
- The Efficiency of Radiation Shielding Sheet to Reduce Radiation Exposure during C-arm Fluoroscopy
- [Korean]
- Recovery and Synthesis of Silver Nanoparticles from Leaching Solution of LTCC Electrode By-Products
- Juyeon Yoo, Yubin Kang, Jinju Park, Hojin Ryu, Jin-Ho Yoon, Kun-Jae Lee
- J Korean Powder Metall Inst. 2017;24(4):315-320. Published online August 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.4.315

- 618 View
- 2 Download
-
 Abstract
Abstract
 PDF
PDF There has been much interest in recycling electronic wastes in order to mitigate environmental problems and to recover the large amount of constituent metals. Silver recovery from electronic waste is extensively studied because of environmental and economic benefits and the use of silver in fabricating nanodevices. Hydrometallurgical processing is often used for silver recovery because it has the advantages of low cost and ease of control. Research on synthesis recovered silver into nanoparticles is needed for application to transistors and solar cells. In this study, silver is selectively recovered from the by-product of electrodes. Silver precursors are prepared using the dissolution characteristics of the leaching solution. In the liquid reduction process, silver nanoparticles are synthesized under various surfactant conditions and then analyzed. The purity of the recovered silver is 99.24%, and the average particle size of the silver nanoparticles is 68 nm.
- [Korean]
- Electrochemical Behavior of Well-dispersed Catalysts on Ruthenium Oxide Nanofiber Supports
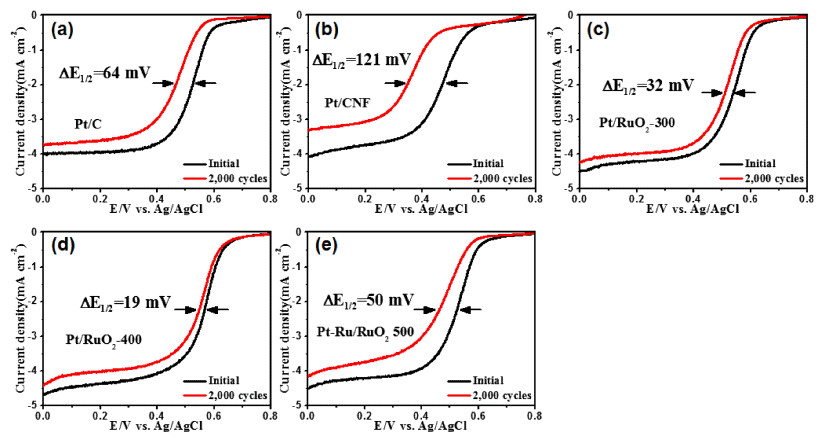
- Geon-Hyoung An, Hyo-Jin Ahn
- J Korean Powder Metall Inst. 2017;24(2):96-101. Published online April 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.2.96
- 1,163 View
- 3 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF Well-dispersed platinum catalysts on ruthenium oxide nanofiber supports are fabricated using electrospinning, post-calcination, and reduction methods. To obtain the well-dispersed platinum catalysts, the surface of the nanofiber supports is modified using post-calcination. The structures, morphologies, crystal structures, chemical bonding energies, and electrochemical performance of the catalysts are investigated. The optimized catalysts show well-dispersed platinum nanoparticles (1-2 nm) on the nanofiber supports as well as a uniform network structure. In particular, the well-dispersed platinum catalysts on the ruthenium oxide nanofiber supports display excellent catalytic activity for oxygen reduction reactions with a half-wave potential (E1/2) of 0.57 V and outstanding long-term stability after 2000 cycles, resulting in a lower E1/2 potential degradation of 19 mV. The enhanced electrochemical performance for oxygen reduction reactions results from the well-dispersed platinum catalysts and unique nanofiber supports.
-
Citations
Citations to this article as recorded by- Fabrication of Porous Electrodes for Zinc-Ion Supercapacitors with Improved Energy Storage Performance
Geon-Hyoung An
Korean Journal of Materials Research.2019; 29(8): 505. CrossRef - Surface tailoring of zinc electrodes for energy storage devices with high-energy densities and long cycle life
Geon-Hyoung An, SeungNam Cha, Jung Inn Sohn
Applied Surface Science.2019; 467-468: 1157. CrossRef - Synthesis of Nitrogen Doped Protein Based Carbon as Pt Catalysts Supports for Oxygen Reduction Reaction
Young-geun Lee, Geon-hyeong An, Hyo-Jin Ahn
Korean Journal of Materials Research.2018; 28(3): 182. CrossRef
- Fabrication of Porous Electrodes for Zinc-Ion Supercapacitors with Improved Energy Storage Performance
- [Korean]
- The Preparation and Growth Mechanism of the Recovered Bi2Te3 Particles with Respect to Surfactants
- Hyeongsub So, Eunpil Song, Yong-Ho Choa, Kun-Jae Lee
- J Korean Powder Metall Inst. 2017;24(2):141-146. Published online April 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.2.141

- 1,410 View
- 9 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF Bi2Te3 powders are recovered by wet chemical reduction for waste n-type thermoelectric chips, and the recovered particles with different morphologies are prepared using various surfactants such as cetyltrimethylammonium bromide (CTAB), sodium dodecylbenzenesulfonate (SDBS), and ethylenediaminetetraacetic acid (EDTA). When citric acid is added as the surfactant, the shape of the aggregated particles shows no distinctive features. On the other hand, rod-shaped particles are formed in the sample with CTAB, and sheet-like particles are synthesized with the addition of SDBS. Further, particles with a tripod shape are observed when EDTA is added as the surfactant. The growth mechanism of the particle shapes depending on the surfactant is investigated, with a focus on the nucleation and growth phenomena. These results help to elucidate the intrinsic formation mechanism of the rod, plate, and tripod structures of the Bi2Te3 recovered by the wet reduction process.
-
Citations
Citations to this article as recorded by- Recovery of Barium, Nickel, and Titanium Powders from Waste MLCC
Haein Shin, Kun-Jae Lee
Journal of Powder Materials.2024; 31(5): 374. CrossRef
- Recovery of Barium, Nickel, and Titanium Powders from Waste MLCC
- [Korean]
- Effect of Heat Treatment Temperature and Atmosphere on the Microstructure of TiH2-WO3 Powder Mixtures
- Han-Eol Lee, Yeon Su Kim, Sung-Tag Oh
- J Korean Powder Metall Inst. 2017;24(1):41-45. Published online February 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.1.41

- 1,120 View
- 2 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF The effects of the heat treatment temperature and of the atmosphere on the dehydrogenation and hydrogen reduction of ball-milled TiH2-WO3 powder mixtures are investigated for the synthesis of Ti-W powders with controlled microstructure. Homogeneously mixed powders with refined TiH2 particles are successfully prepared by ball milling for 24 h. X-ray diffraction (XRD) analyses show that the powder mixture heat-treated in Ar atmosphere is composed of Ti, Ti2O, and W phases, regardless of the heat treatment temperature. However, XRD results for the powder mixture, heat-treated at 600°C in a hydrogen atmosphere, show TiH2 and TiH peaks as well as reaction phase peaks of Ti oxides and W, while the powder mixture heat-treated at 900°C exhibits only XRD peaks attributed to Ti oxides and W. The formation behavior of the reaction phases that are dependent on the heat treatment temperature and on the atmosphere is explained by thermodynamic considerations for the dehydrogenation reaction of TiH2, the hydrogen reduction of WO3 and the partial oxidation of dehydrogenated Ti.
-
Citations
Citations to this article as recorded by- Fabrication of Porous W-Ti by Freeze-Drying and Hydrogen Reduction of WO3-TiH2 Powder Mixtures
Hyunji Kang, Sung Hyun Park, Sung-Tag Oh
Journal of Korean Powder Metallurgy Institute.2017; 24(6): 472. CrossRef
- Fabrication of Porous W-Ti by Freeze-Drying and Hydrogen Reduction of WO3-TiH2 Powder Mixtures
- [Korean]
- Basic Study on the Recycling of Waste Tungsten Scraps by the Oxidation and Reduction Process
- Sang-Uk Kim, Ji-seok Yun, Tae-Wook Kim, Bong-Hwi Cho, In-Ho Kim, Sang-Mu Kim, Chang-Bin Song
- J Korean Powder Metall Inst. 2017;24(1):34-40. Published online February 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.1.34

- 1,017 View
- 4 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF This study is carried out to obtain basic data regarding oxidation and reduction reactions, originated on the recycling of waste tungsten hard scraps by oxidation and reduction processes. First, it is estimated that the theoretical Gibbs free energy for the formation reaction of WO2 and WO3 are calculated as ΔG1,000K= -407.335 kJ/mol and ΔG1,000K = -585.679 kJ/mol, from the thermodynamics data reported by Ihsan Barin. In the experiments, the oxidation of pure tungsten rod by oxygen is carried out over a temperature range of 700-1,000°C for 1 h, and it is possible to conclude that the oxidation reaction can be represented by a relatively linear relationship. Second, the reduction of WO2 and WO3 powder by hydrogen is also calculated from the same thermodynamics data, and it can be found that it was difficult for the reduction reaction to occur at 1,027°C, in the case of WO2, but it can happen for temperatures higher than 1127°C. On the other hand, WO3 reduction reaction occurs at the relatively low temperature of 827oC. Based on these results, the reduction experiments are carried out at a temperature range of 500-1,000°C for 15 min to 4 h, in the case of WO3 powder, and it is possible to conclude that the reduction at 900°C for 2h is needed for a perfect reduction reaction.
-
Citations
Citations to this article as recorded by- A Basic Study on the Recycling of Wasted Cemented Carbide by the Zn Bath Process(Ⅰ)
Kyung-Sik Kim, In-Ho Kim, Chan-Gi Lee, Chang-Bin Song
Journal of the Korean Institute of Resources Recycling.2020; 29(6): 35. CrossRef
- A Basic Study on the Recycling of Wasted Cemented Carbide by the Zn Bath Process(Ⅰ)
- [Korean]
- Synthesis of Nitrogen-doped Carbon Nanofibers for Oxygen Reduction Reaction
- Geon-Hyoung An, Eun-Hwan Lee, Hyo-Jin Ahn
- J Korean Powder Metall Inst. 2016;23(6):420-425. Published online December 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.6.420

- 870 View
- 2 Download
-
 Abstract
Abstract
 PDF
PDF N-doped carbon nanofibers as catalysts for oxygen-reduction reactions are synthesized using electrospinning and carbonization. Their morphologies, structures, chemical bonding states, and electrochemical performance are characterized. The optimized N-doped carbon nanofibers exhibit graphitization of carbon nanofibers and an increased nitrogen doping as well as a uniform network structure. In particular, the optimized N-doped carbon nanofibers show outstanding catalytic activity for oxygen-reduction reactions, such as a half-wave potential (E1/2) of 0.43 V, kinetic limiting current density of 6.2 mA cm-2, electron reduction pathways (n = 3.1), and excellent long-term stability after 2000 cycles, resulting in a lower E1/2 potential degradation of 13 mV. The improvement in the electrochemical performance results from the synergistic effect of the graphitization of carbon nanofibers and the increased amount of nitrogen doping.
- [Korean]
- Effect of Heat Treatment Atmosphere on the Microstructure of TiH2-MoO3 Powder Mixtures
- Ki Cheol Jeon, Sung Hyun Park, Na-Yeon Kwon, Sung-Tag Oh
- J Korean Powder Metall Inst. 2016;23(4):303-306. Published online August 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.4.303

- 1,206 View
- 3 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF An optimum route to synthesize Ti-Mo system powders is investigated by analyzing the effect of the heat treatment atmosphere on the formation of the reaction phase by dehydrogenation and hydrogen reduction of ball-milled TiH2-MoO3 powder mixtures. Homogeneous powder mixtures with refined particles are prepared by ball milling for 24 h. XRD analysis of the heat-treated powder in a hydrogen atmosphere shows TiH2 and MoO3 peaks in the initial powders as well as the peaks corresponding to the reaction phase species, such as TiH0.7, TiO, MoO2, Mo. In contrast, powder mixtures heated in an argon atmosphere are composed of Ti, TiO, Mo and MoO3 phases. The formation of reaction phases dependent on the atmosphere is explained by the partial pressure of H2 and the reaction temperature, based on thermodynamic considerations for the dehydrogenation reaction of TiH2 and the reduction behavior of MoO3.
-
Citations
Citations to this article as recorded by- Effect of titanium addition on mechanical properties of Mo-Si-B alloys
Won June Choi, Seung Yeong Lee, Chun Woong Park, Jung Hyo Park, Jong Min Byun, Young Do Kim
International Journal of Refractory Metals and Hard Materials.2019; 80: 238. CrossRef
- Effect of titanium addition on mechanical properties of Mo-Si-B alloys
- [Korean]
- Fabrication and Characterization of Ag Particles by Polyol Process and Wet Chemical Process
- Juyeon Yoo, Hyosung Jang, Kun-Jae Lee
- J Korean Powder Metall Inst. 2016;23(4):297-302. Published online August 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.4.297
- 1,053 View
- 3 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF Ag nanoparticles are extensively studied and utilized due to their excellent catalysis, antibiosis and optical properties. They can be easily synthesized by chemical reduction methods and it is possible to prepare particles of uniform size and high purity. These methods are divided into vapor methods and liquid phase reduction methods. In the present study, Ag particles are prepared and analyzed through two chemical reduction methods using solvents containing a silver nitrate precursor. When Ag ions are reduced using a reductant in the aqueous solution, it is possible to control the Ag particle size by controlling the formic acid ratio. In addition, in the Polyol process, Ag nanoparticles prepared at various temperatures and reaction time conditions have multiple twinned and anisotropic structures, and the particle size variation can be confirmed using field emissions scanning electron microscopy and by analyzing the UV-vis spectrum.
-
Citations
Citations to this article as recorded by- Recovery and Synthesis of Silver Nanoparticles from Leaching Solution of LTCC Electrode By-Products
Juyeon Yoo, Yubin Kang, Jinju Park, Hojin Ryu, Jin-Ho Yoon, Kun-Jae Lee
Journal of Korean Powder Metallurgy Institute.2017; 24(4): 315. CrossRef
- Recovery and Synthesis of Silver Nanoparticles from Leaching Solution of LTCC Electrode By-Products
- [Korean]
- Synthesis of Copper Nanoparticles by a Chemical Reduction Method
- Min Woo Choi, Min Hwan Bae, Jung-Ho Ahn
- J Korean Powder Metall Inst. 2016;23(3):228-234. Published online June 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.3.228

- 2,630 View
- 50 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF Copper nanoparticles attract much attention as substitutes of noble metals such as silver and can help reduce the manufacturing cost of electronic products due to their lower cost and good conductivity. In the present work, the chemical reduction is examined to optimize the synthesis of nano-sized copper particles from copper sulfate. Sodium borohydride and ascorbic acid are used as reducing and antioxidant agents, respectively. Polyethylene glycol (PEG) is used as a size-control and capping agent. An appropriate dose of PEG inhibits the abnormal growth of copper nanoparticles, maintaining chemical stability. The addition of ascorbic acid prevents the oxidation of nanoparticles during synthesis and storage. Transmission electron microscopy (TEM) and Fourier transform infrared spectroscopy (FTIR) are used to investigate the size of the synthesized nanoparticles and the coordination between copper nanoparticles and PEG. For chemical reduction, copper nanoparticles less than 100 nm in size without oxidized layers are successfully obtained by the present method.
-
Citations
Citations to this article as recorded by- Correlation between electrical conductivity and antibacterial activity of chitosan-stabilized copper and silver nanoparticles
C.Raja Mohan, Ruckmani Kandasamy, J. Kabiriyel
Carbohydrate Polymer Technologies and Applications.2024; 7: 100503. CrossRef - Green Synthesis and Characterization of Natural Magnetic Particles/Chitosan Composite Material Impregnated with Copper Nanoparticles
Defia Indah Permatasari, Bambang Rusdiarso, Nuryono Nuryono
Solid State Phenomena.2022; 339: 19. CrossRef - Copper Nanoparticle(CuNP’s)Synthesis: A review of the various ways with Photocatalytic and Antibacterial Activity
Israfil Alam Tito, Sahab Uddin, Shafiul Islam, Snahasish Bhowmik
Oriental Journal Of Chemistry.2021; 37(5): 1030. CrossRef
- Correlation between electrical conductivity and antibacterial activity of chitosan-stabilized copper and silver nanoparticles
- [English]
- Effects of Hydrogen Reduction in Microstructure, Mechanical and Thermoelectric Properties of Gas Atomized
n -type Bi2Te2.7 Se0.3 Material - Pradip Rimal, Sang-Min Yoon, Eun-Bin Kim, Chul-Hee Lee, Soon-Jik Hong
- J Korean Powder Metall Inst. 2016;23(2):126-131. Published online April 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.2.126

- 1,266 View
- 5 Download
- 6 Citations
-
 Abstract
Abstract
 PDF
PDF The recent rise in applications of thermoelectric materials has attracted interest in studies toward the fabrication of thermoelectric materials using mass production techniques. In this study, we successfully fabricate
n -type Bi2Te2.7Se0.3 material by a combination of mass production powder metallurgy techniques, gas atomization, and spark plasma sintering. In addition, to examine the effects of hydrogen reduction in the microstructure, the thermoelectric and mechanical properties are measured and analyzed. Here, almost 60% of the oxygen content of the powder are eliminated after hydrogen reduction for 4 h at 360°C. Micrographs of the powder show that the reduced powder had a comparatively clean surface and larger grain sizes than unreduced powder. The density of the consolidated bulk using as-atomized powder and reduced atomized powder exceeds 99%. The thermoelectric power factor of the sample prepared by reduction of powder is 20% better than that of the sample prepared using unreduced powder.-
Citations
Citations to this article as recorded by- Tuning of power factor in bismuth selenide through Sn/Te co doping for low temperature thermoelectric applications
Ganesh Shridhar Hegde, Ashwatha Narayana Prabhu, Ramakrishna Nayak, C. F. Yang, Y. K. Kuo
Applied Physics A.2024;[Epub] CrossRef - Enhancing thermoelectric performance of K-doped polycrystalline SnSe through band engineering tuning and hydrogen reduction
Nan Xin, Yifei Li, Guihua Tang, Longyun Shen
Journal of Alloys and Compounds.2022; 899: 163358. CrossRef - The effect of powder pre-treatment on the mechanical and thermoelectric properties of spark plasma sintered N-type bismuth telluride
Ahmed A. Abdelnabi, Vickram Lakhian, Joseph R. McDermid, James S. Cotton
Journal of Alloys and Compounds.2021; 874: 159782. CrossRef - Investigation of Spark Plasma Sintering Temperature on Microstructure and Thermoelectric Properties of p-type Bi-Sb-Te alloys
Jin-Koo Han, Dong-won Shin, Babu Madavali, Soon-Jik Hong
Journal of Korean Powder Metallurgy Institute.2017; 24(2): 115. CrossRef - The Preparation and Growth Mechanism of the Recovered Bi2Te3 Particles with Respect to Surfactants
Hyeongsub So, Eunpil Song, Yong-Ho Choa, Kun-Jae Lee
Journal of Korean Powder Metallurgy Institute.2017; 24(2): 141. CrossRef - Enhanced thermoelectric cooling properties of Bi2Te3−xSex alloys fabricated by combining casting, milling and spark plasma sintering
Seung Tek Han, Pradip Rimal, Chul Hee Lee, Hyo-Seob Kim, Yongho Sohn, Soon-Jik Hong
Intermetallics.2016; 78: 42. CrossRef
- Tuning of power factor in bismuth selenide through Sn/Te co doping for low temperature thermoelectric applications
- [Korean]
- Effect of Freezing and Sintering Condition of CuO-SnO2/Camphene Slurries on the Pore Structure of Porous Cu-Sn
- Joo-Hyung Kim, Sung-Tag Oh, Chang-Yong Hyun
- J Korean Powder Metall Inst. 2016;23(1):49-53. Published online February 1, 2016
- DOI: https://doi.org/10.4150/KPMI.2016.23.1.49

- 881 View
- 1 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF The present study demonstrates the effect of freezing conditions on the pore structure of porous Cu-10 wt.% Sn prepared by freeze drying of CuO-SnO2/camphene slurry. Mixtures of CuO and SnO2 powders are prepared by ball milling for 10 h. Camphene slurries with 10 vol.% of CuO-SnO2 are unidirectionally frozen in a mold maintained at a temperature of -30°C for 1 and 24 h, respectively. Pores are generated by the sublimation of camphene at room temperature. After hydrogen reduction and sintering at 650°C for 2 h, the green body of the CuO-SnO2 is completely converted into porous Cu-Sn alloy. Microstructural observation reveals that the sintered samples have large pores which are aligned parallel to the camphene growth direction. The size of the large pores increases from 150 to 300 μm with an increase in the holding time. Also, the internal walls of the large pores contain relatively small pores whose size increases with the holding time. The change in pore structure is explained by the growth behavior of the camphene crystals and rearrangement of the solid particles during the freezing process.
-
Citations
Citations to this article as recorded by- Fabrication of Porous Ni by Freeze Drying and Hydrogen Reduction of NiO/Camphene Slurry
Jae-Hun Jeong, Sung-Tag Oh, Chang-Yong Hyun
Journal of Korean Powder Metallurgy Institute.2019; 26(1): 6. CrossRef - Fabrication of Al2O3 Dispersed Porous Cu by Freeze Drying of CuO-Al2O3/Camphene Slurry
Hyunji Kang, Doh-Hyung Riu, Sung-Tag Oh
journal of Korean Powder Metallurgy Institute.2018; 25(1): 25. CrossRef - Porous W-Ni Alloys Synthesized from Camphene/WO3-NiO Slurry by Freeze Drying and Heat Treatment in Hydrogen Atmosphere
Sung Hyun Park, Seong-Min Park, So-Jeong Park, Bo-Yeong Park, Sung-Tag Oh
Korean Journal of Materials Research.2018; 28(2): 108. CrossRef
- Fabrication of Porous Ni by Freeze Drying and Hydrogen Reduction of NiO/Camphene Slurry
- [Korean]
- Synthesis of NiTi Alloy Powder by the Reaction of NiO-TiH2 Mixing Powders
- Ki Cheol Jeon, Han-Eol Lee, Da-Mi Yim, Sung-Tag Oh
- J Korean Powder Metall Inst. 2015;22(4):266-270. Published online August 1, 2015
- DOI: https://doi.org/10.4150/KPMI.2015.22.4.266

- 1,068 View
- 2 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF The synthesis of NiTi alloy powders by hydrogen reduction and dehydrogenation process of NiO and TiH2 powder mixtures is investigated. Mixtures of NiO and TiH2 powders are prepared by simple mixing for 1 h or ball milling for 24 h. Simple-mixed mixture shows that fine NiO particles are homogeneously coated on the surface of TiH2 powders, whereas ball milled one exhibits the morphology with mixing of fine NiO and TiH2 particles. Thermogravimetric analysis in hydrogen atmosphere reveals that the NiO and TiH2 phase are changed to metallic Ni and Ti in the temperature range of 260 to 290°C and 553 to 639°C, respectively. In the simple-mixed powders by heat-up to 700°C, agglomerates with solid particles and solidified liquid phase are observed, and the size of agglomerates is increased at 1000°C. From the XRD analysis, the presence of liquid phase is explained by the formation and melting of NiTi2 intermetallic compound due to an exothermic reaction between Ni and Ti. The simple-mixed powders, heated to 1000°C, lead to the formation of NiTi phase but additional Ni-, Ti-rich and Ti-oxide phases. In contrast, the microstructure of ball-milled powders is characterized by the neck-grown particles, forming Ni3Ti, Ti-oxide and unreacted Ni phase.
-
Citations
Citations to this article as recorded by- Effect of Heat Treatment Atmosphere on the Microstructure of TiH2-MoO3 Powder Mixtures
Ki Cheol Jeon, Sung Hyun Park, Na-Yeon Kwon, Sung-Tag Oh
Journal of Korean Powder Metallurgy Institute.2016; 23(4): 303. CrossRef
- Effect of Heat Treatment Atmosphere on the Microstructure of TiH2-MoO3 Powder Mixtures
- [Korean]
- Preparation of Spherical Cobalt Fine Powders by New Liquid Reduction Method
- Dae Weon Kim, Ji-Hoon Kim, Yo-Han Choi, Hee Lack Choi, Jin-Ho Yoon
- J Korean Powder Metall Inst. 2015;22(4):260-265. Published online August 1, 2015
- DOI: https://doi.org/10.4150/KPMI.2015.22.4.260
- 1,009 View
- 5 Download
-
 Abstract
Abstract
 PDF
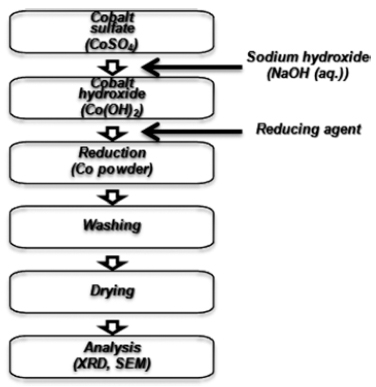
PDF Spherical fine cobalt powders were fabricated by new liquid reduction method. Commercial cobalt sufate heptahydrate was used as raw material. Also ethylene glycol was used as solvent and hydrazine-sodium hypophosphite mixture was used as reduction agent for the new liquid reduction method. A plate shaped cobalt powders with an approximately 300 nm were prepared by a traditional wet ruduction method using distilled water as solvent and hydrazine. Spherical fine cobalt powders with an average size of 1-3 μm were synthesized by a new liquid reduction method in 0.3M cobalt sulfate and 1.5M hydrazine-0.6M sodium hypophosphite mixture at 333K.
- [Korean]
- Fabrication of Porous W by Heat Treatment of Pore Forming Agent of PMMA and WO3 Powder Compacts
- Ki Cheol Jeon, Young Do Kim, Myung-Jin Suk, Sung-Tag Oh
- J Korean Powder Metall Inst. 2015;22(2):129-133. Published online April 1, 2015
- DOI: https://doi.org/10.4150/KPMI.2015.22.2.129

- 1,993 View
- 7 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF Porous W with controlled pore structure was fabricated by thermal decomposition and hydrogen reduction process of PMMA beads and WO3 powder compacts. The PMMA sizes of 8 and 50 μm were used as pore forming agent for fabricating the porous W. The WO3 powder compacts with 20 and 70 vol% PMMA were prepared by uniaxial pressing and sintered for 2 h at 1200°C in hydrogen atmosphere. TGA analysis revealed that the PMMA was decomposed at about 400°C and WO3 was reduced to metallic W at 800°C. Large pores in the sintered specimens were formed by thermal decomposition of spherical PMMA, and their size was increased with increase in PMMA size and the amount of PMMA addition. Also the pore shape was changed from spherical to irregular form with increasing PMMA contents due to the agglomeration of PMMA in the powder mixing process.
-
Citations
Citations to this article as recorded by- Synthesis of Porous Silica Particles Using Sodium Silicate Precursor for Water-Repellent Surfaces
Young-Sang Cho, Nahee Ku, Young-Seok Kim
JOURNAL OF CHEMICAL ENGINEERING OF JAPAN.2019; 52(2): 194. CrossRef
- Synthesis of Porous Silica Particles Using Sodium Silicate Precursor for Water-Repellent Surfaces
- [Korean]
- Effect of Oxygen Content in the Tungsten Powder Fabricated by Electrical Explosion of Wire Method on the Behavior of Spark-Plasma Sintering
- Cheol-Hee Kim, Seong Lee, Byung-Kee Kim, Ji Soon Kim
- J Korean Powder Metall Inst. 2014;21(6):447-453. Published online December 1, 2014
- DOI: https://doi.org/10.4150/KPMI.2014.21.6.447
- 848 View
- 4 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
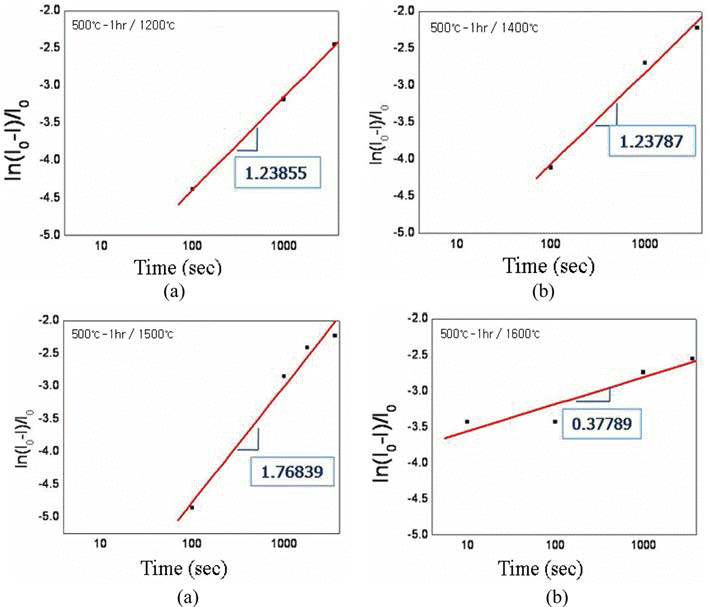
PDF Effect of oxygen content in the ultrafine tungsten powder fabricated by electrical explosion of wire method on the behvior of spark plasma sintering was investigated. The initial oxygen content of 6.5 wt% of as-fabricated tungsten powder was reduced to 2.3 and 0.7 wt% for the powders which were reduction-treated at 400°C for 2 hour and at 500°C for 1h in hydrogen atmosphere, respectively. The reduction-treated tungsten powders were spark-plasma sintered at 1200-1600°C for 100-3600 sec. with applied pressure of 50 MPa under vacuum of 0.133 Pa. Maximun sindered density of 97% relative density was obtained under the condition of 1600°C for 1h from the tungsten powder with 0.7 wt% oxygen. Sintering activation energy of 95.85 kJ/mol−1 was obtained, which is remarkably smaller than the reported ones of 380~460 kJ/mol−1 for pressureless sintering of micron-scale tungsten powders.
-
Citations
Citations to this article as recorded by- Effect of Powder Mixing Process on the Characteristics of Hybrid Structure Tungsten Powders with Nano-Micro Size
Na-Yeon Kwon, Young-Keun Jeong, Sung-Tag Oh
Journal of Korean Powder Metallurgy Institute.2017; 24(5): 384. CrossRef
- Effect of Powder Mixing Process on the Characteristics of Hybrid Structure Tungsten Powders with Nano-Micro Size
- [Korean]
- Synthesis of Silica-Core Gold-Satellite Nanoparticles and Their Surface-enhanced Raman Scattering Based Sensing Application
- Hyun Ji Choi, Young-Kuk Kim, Seok-Young Yoon, Youn-Kyoung Baek
- J Korean Powder Metall Inst. 2014;21(6):441-446. Published online December 1, 2014
- DOI: https://doi.org/10.4150/KPMI.2014.21.6.441
- 1,705 View
- 7 Download
-
 Abstract
Abstract
 PDF
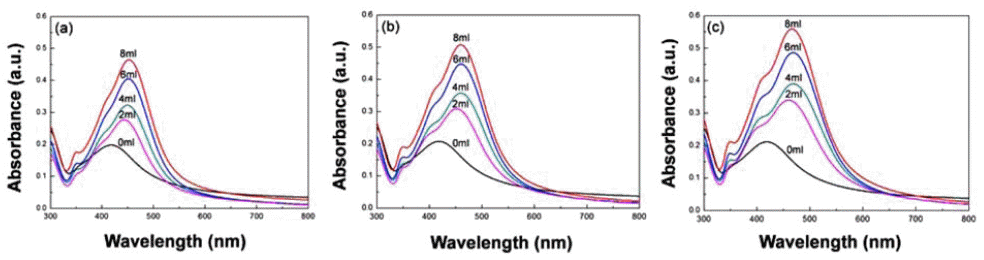
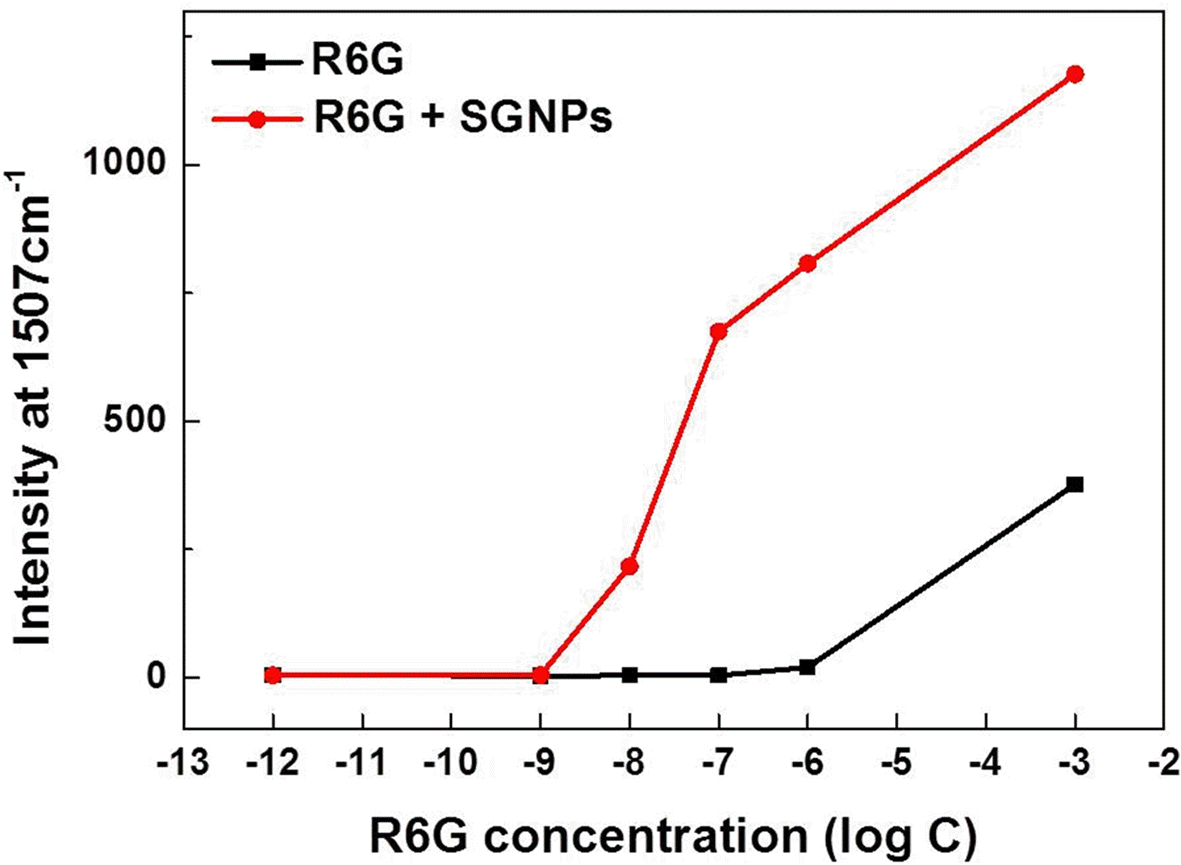
PDF In this study, we synthesize silica-core gold-satellite nanoparticles (SGNPs) for the surface-enhanced Raman scattering (SERS) based sensing applications. They consist of gold satellite nanoparticles (AuNPs) fixed on the silica core nanoparticles, which sizes of AuNPs can be tunned by varying the amount of reactants (growth solution and reducing agent). Their surface plasmon resonance (SPR) properties were characterized by using UV-vis spectroscopy, showing that the growth of AuNPs on silica cores leads to the light absorption in the longer wavelength region. Furthermore, the size increase of AuNPs exhibited the dramatic change in SERS activity due to the formation of hot spots. The optimized SGNPs showing enhancement factor~3.8×106 exhibited a detection limit of rhodamine 6G (R6G) as low as 10−8 M. These findings suggest the importance of size control of SGNPs and their SPR properties to develop highly efficient SERS sensors.
- [Korean]
- Hydrogen Reduction Behavior of Oxide Scale in Water-atomized Iron Powder
- Hea-Min Shin, Kyeong-Ho Baik
- J Korean Powder Metall Inst. 2014;21(6):422-428. Published online December 1, 2014
- DOI: https://doi.org/10.4150/KPMI.2014.21.6.422
- 1,458 View
- 22 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF In this study, the reduction kinetics and behaviors of oxides in the water-atomized iron powder have been evaluated as a function of temperature ranging 850-1000°C in hydrogen environment, and compared to the reduction behaviors of individual iron oxides including Fe2O3, Fe3O4 and FeO. The water-atomized iron powder contained a significant amount of iron oxides, mainly Fe3O4 and FeO, which were formed as a partially-continuous surface layer and an inner inclusion. During hydrogen reduction, a significant weight loss in the iron powder occurred in the initial stage of 10 min by the reduction of surface oxides, and then further reduction underwent slowly with increasing time. A higher temperature in the hydrogen reduction promoted a high purity of iron powder, but no significant change in the reduction occurred above 950°C. Sequence reduction process by an alternating environment of hydrogen and inert gases effectively removed the oxide scale in the iron powder, which lowered reduction temperature and/or shortened reduction time.
-
Citations
Citations to this article as recorded by- Carbon Co-Deposition During Gas Reduction of Water-Atomized Fe-Cr-Mo Powder
B. Ali, S.H. Choi, S.J. Seo, D.Y. Maeng, C.G. Lee, T.S. Kim, K.T. Park
Archives of Metallurgy and Materials.2017; 62(2): 1119. CrossRef
- Carbon Co-Deposition During Gas Reduction of Water-Atomized Fe-Cr-Mo Powder
- [Korean]
- Analysis of the Change in Microstructures of Nano Copper Powders During the Hydrogen Reduction using X-ray Diffraction Patterns and Transmission Electron Microscope, and the Mechanical Property of Compacted Powders
- Dong-Hyun Ahn, Dong Jun Lee, Wooyeol Kim, Lee Ju Park, Hyoung Seop Kim
- J Korean Powder Metall Inst. 2014;21(3):207-214. Published online June 1, 2014
- DOI: https://doi.org/10.4150/KPMI.2014.21.3.207

- 789 View
- 2 Download
-
 Abstract
Abstract
 PDF
PDF In this study, nano-scale copper powders were reduction treated in a hydrogen atmosphere at the relatively high temperature of 350°C in order to eliminate surface oxide layers, which are the main obstacles for fabricating a nano/ultrafine grained bulk parts from the nano-scale powders. The changes in composition and microstructure before and after the hydrogen reduction treatment were evaluated by analyzing X-ray diffraction (XRD) line profile patterns using the convolutional multiple whole profile (CMWP) procedure. In order to confirm the result from the XRD line profile analysis, transmitted electron microscope observations were performed on the specimen of the hydrogen reduction treated powders fabricated using a focused ion beam process. A quasi-statically compacted specimen from the nanoscale powders was produced and Vickers micro-hardness was measured to verify the potential of the powders as the basis for a bulk nano/ultrafine grained material. Although the bonding between particles and the growth in size of the particles occurred, crystallites retained their nano-scale size evaluated using the XRD results. The hardness results demonstrate the usefulness of the powders for a nano/ultrafine grained material, once a good consolidation of powders is achieved.
- [English]
- Coating of Cobalt Over Tungsten Carbide Powder by Wet Chemical Reduction Method
- Hyun-Seon Hong, Jin-Ho Yoon
- J Korean Powder Metall Inst. 2014;21(2):93-96. Published online April 1, 2014
- DOI: https://doi.org/10.4150/KPMI.2014.21.2.93
- 2,202 View
- 9 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF Cobalt coated tungsten carbide-cobalt composite powder has been prepared through wet chemical reduction method. The cobalt sulfate solution was converted to the cobalt chloride then the cobalt hydroxide. The tungsten carbide powders were added in to the cobalt hydroxide, the cobalt hydroxide was reduced and coated over tungsten carbide powder using hypo-phosphorous acid. Both the cobalt and the tungsten carbide phase peaks were evident in the tungsten carbide-cobalt composite powder by X-ray diffraction. The average particle size measured via scanning electron microscope, particle size analysis was around 380 nm and the thickness of coated cobalt was determined to be 30~40 nm by transmission electron microscopy.
-
Citations
Citations to this article as recorded by- Electroless Ni-P deposition on WC powders through direct PdCl2 activation and study on the underlying mechanisms
Peng Tang, Shuwen Jiang, Jiawei Yan, Xianquan Li
Next Materials.2025; 6: 100496. CrossRef - Pre-treatments of initial materials for controlling synthesized TaC characteristics in the SHS process
Jae Jin Sim, Sang Hoon Choi, Ji Hwan Park, Il Kyu Park, Jae Hong Lim, Kyoung Tae Park
journal of Korean Powder Metallurgy Institute.2018; 25(3): 251. CrossRef - Spark plasma sintering of WC–Co tool materials prepared with emphasis on WC core–Co shell structure development
Sungkyu Lee, Hyun Seon Hong, Hyo-Seob Kim, Soon-Jik Hong, Jin-Ho Yoon
International Journal of Refractory Metals and Hard Materials.2015; 53: 41. CrossRef
- Electroless Ni-P deposition on WC powders through direct PdCl2 activation and study on the underlying mechanisms
- [Korean]
- Fabrication of Porous Cu-Ni by Freeze Drying and Hydrogen Reduction of CuO-NiO Powder Mixture
- Han Gil Seo, Sung-Tag Oh
- J Korean Powder Metall Inst. 2014;21(1):34-38. Published online February 1, 2014
- DOI: https://doi.org/10.4150/KPMI.2014.21.1.34

- 1,197 View
- 0 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF Cu-Ni alloys with unidirectionally aligned pores were prepared by freeze-drying process of CuO-NiO/camphene slurry. Camphene slurries with dispersion stability by the addition of oligomeric polyester were frozen at -25°C, and pores in the frozen specimens were generated by sublimation of the camphene during drying in air. The green bodies were hydrogen-reduced at 300°C and sintered at 850°C for 1 h. X-ray diffraction analysis revealed that CuO-NiO composite powders were completely converted to Cu-Ni alloy without any reaction phases by hydrogen reduction. The sintered samples showed large and aligned parallel pores to the camphene growth direction, and small pores in the internal wall of large pores. The pore size and porosity decreased with increase in CuO-NiO content from 5 to 10 vol%. The change of pore characteristics was explained by the degree of powder rearrangement in slurry and the accumulation behavior of powders in the interdendritic spaces of solidified camphene.
-
Citations
Citations to this article as recorded by- Enhancement in electrical conductivity of pastes containing submicron Ag-coated Cu filler with palmitic acid surface modification
Eun Byeol Choi, Jong-Hyun Lee
Applied Surface Science.2017; 415: 67. CrossRef - Investigation for Microstructure and Hardness of Welded Zone of Cu-Ni Alloy using W92-Ni-Fe Sintering Tool
Tae-Jin Yoon, Sang-Won Park, Myung-Chang Kang, Joong-Suk Noh, Sung-Wook Chung, Chung-Yun Kang
Journal of Korean Powder Metallurgy Institute.2015; 22(3): 181. CrossRef - Controlling Structural and Electrical Properties of Pt Nanopowder-Dispersed SiO2Film
Jae Ho Lee, In Joo Shin, Sung Woo Lee, Hyeong Cheol Kim, Byung Joon Choi
Journal of Korean Powder Metallurgy Institute.2014; 21(5): 355. CrossRef
- Enhancement in electrical conductivity of pastes containing submicron Ag-coated Cu filler with palmitic acid surface modification
TOP
 KPMI
KPMI


 First
First Prev
Prev


