Search
- Page Path
- HOME > Search
- [Korean]
- High-Hardness Cemented Carbide With Nickel-Tungsten Alloy Binder
- Hanjung Kwon
- J Powder Mater. 2024;31(4):318-323. Published online August 30, 2024
- DOI: https://doi.org/10.4150/jpm.2024.00227
- 1,137 View
- 28 Download
-
 Abstract
Abstract
 PDF
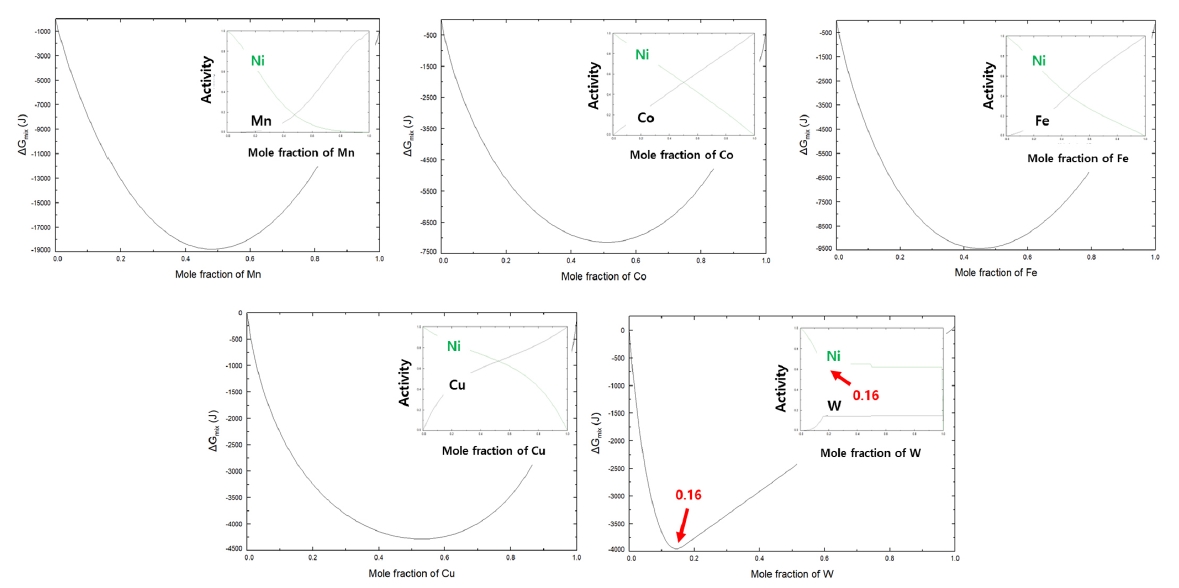
PDF - Cemented carbide for cutting tools, which is composed of carbide as a hard phase and metallic component as a metallic phase, mainly uses cobalt as the metallic phase due to the excellent mechanical properties of cobalt. However, as the demand for machining difficult-to-machine materials such as titanium and carbon fiber-reinforced plastics has recently increased, the development of high-hardness cemented carbide is necessary and the replacement of cobalt metal with a high-hardness alloy is required. In this study, we would like to introduce high-hardness cemented carbide fabricated using nickel-tungsten alloy as the metallic phase. First, nickel-tungsten alloy powder of the composition for formation of intermetallic compound confirmed through thermodynamic calculations was synthesized, and cemented carbide was prepared through the sintering process of tungsten carbide and the synthesized alloy powder. Through evaluating the mechanical properties of high-hardness cemented carbide with the nickel-tungsten alloy binder, the possibility of producing high-hardness cemented carbide by using the alloys with high-hardness was confirmed.
- [Korean]
- Fabrication and Sintering Behavior Analysis of Molybdenum-tungsten Nanopowders by Pechini Process
- Suyeon Kim, Taehyun Kwon, Seulgi Kim, Dongju Lee
- J Powder Mater. 2023;30(5):436-441. Published online October 1, 2023
- DOI: https://doi.org/10.4150/KPMI.2023.30.5.436
- 1,151 View
- 11 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
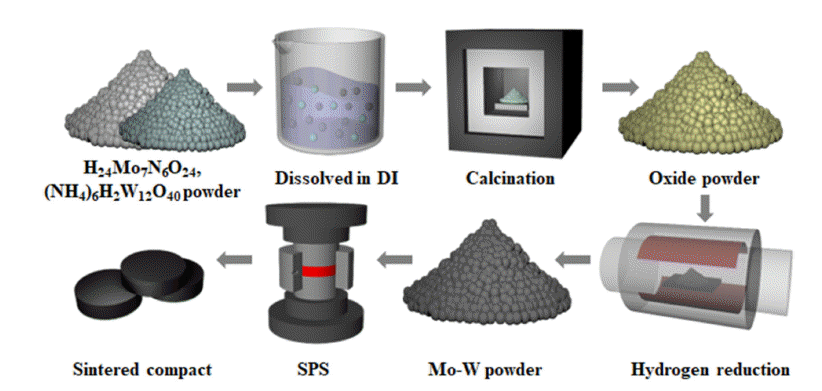
PDF Molybdenum-tungsten (Mo-W) alloy sputtering targets are widely utilized in fields like electronics, nanotechnology, sensors, and as gate electrodes for TFT-LCDs, owing to their superior properties such as hightemperature stability, low thermal expansion coefficient, electrical conductivity, and corrosion resistance. To achieve optimal performance in application, these targets’ purity, relative density, and grain size of these targets must be carefully controlled. We utilized nanopowders, prepared via the Pechini method, to obtain uniform and fine powders, then carried out spark plasma sintering (SPS) to densify these powders. Our studies revealed that the sintered compacts made from these nanopowders exhibited outstanding features, such as a high relative density of more than 99%, consistent grain size of 3.43 μm, and shape, absence of preferred orientation.
-
Citations
Citations to this article as recorded by- Ultrafast Synthesis of Molybdenum Disulfide via Flashlamp Annealing
Chan Hyeon Yang, Jaehak Lee, Jung Hwan Park
Journal of Powder Materials.2025; 32(6): 509. CrossRef
- Ultrafast Synthesis of Molybdenum Disulfide via Flashlamp Annealing
- [Korean]
- Comparison of Properties with Different Sintering Process of 3Y-TZP/WC Composites
- Min-Soo Nam, Jae-Hyung Choi, Sahn Nahm, Seongwon Kim
- J Powder Mater. 2022;29(5):424-431. Published online October 1, 2022
- DOI: https://doi.org/10.4150/KPMI.2022.29.5.424

- 1,059 View
- 4 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF 3Y-TZP ceramics obtained by doping 3 mol.% of Y2O3 to ZrO2 to stabilize the phase transition are widely used in the engineering ceramic industry due to their excellent mechanical properties such as high strength, fracture toughness, and wear resistance. An additional increase in mechanical properties is possible by manufacturing a composite in which a high-hardness material such as oxide or carbide is added to the 3Y-TZP matrix. In this study, composite powder was prepared by dispersing a designated percentage of WC in the 3Y-TZP matrix, and the results were compared after manufacturing the composite using the different processes of spark plasma sintering and HP. The difference between the densification behavior and porosity with the process mechanism was investigated. The correlation between the process conditions and phase formation was examined based on the crystalline phase formation behavior. Changes to the microstructure according to the process conditions were compared using field-emission scanning electron microscopy. The toughness-strengthening mechanism of the composite with densification and phase formation was also investigated.
-
Citations
Citations to this article as recorded by- Phase Formation and Mechanical Property of YSZ‒30 vol.% WC Composite Ceramics Fabricated by Hot Pressing
Jin-Kwon Kim, Jae-Hyeong Choi, Nahm Sahn, Sung-Soo Ryu, Seongwon Kim
journal of Korean Powder Metallurgy Institute.2023; 30(5): 409. CrossRef
- Phase Formation and Mechanical Property of YSZ‒30 vol.% WC Composite Ceramics Fabricated by Hot Pressing
- [Korean]
- A Study on Mechano-chemical Ball Milling Process for Fabricating Tungsten Disulfide Nanosheets
- Seulgi Kim, Yunhee Ahn, Dongju Lee
- J Powder Mater. 2022;29(5):376-381. Published online October 1, 2022
- DOI: https://doi.org/10.4150/KPMI.2022.29.5.376

- 756 View
- 3 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF Tungsten disulfide (WS2) nanosheets have attracted considerable attention because of their unique optical and electrical properties. Several methods for fabrication of WS2 nanosheets have been developed. However, methods for mass production of high-quality WS2 nanosheets remain challenging. In this study, WS2 nanosheets were fabricated using mechano-chemical ball milling based on the synergetic effects of chemical intercalation and mechanical exfoliation. The ball-milling time was set as a variable for the optimized fabricating process of WS2 nanosheets. Under the optimized conditions, the WS2 nanosheets had lateral sizes of 500–600 nm with either a monolayer or bilayer. They also exhibited high crystallinity in the 2H semiconducting phase. Thus, the proposed method can be applied to the exfoliation of other transition metal dichalcogenides using suitable chemical intercalants. It can also be used with highperformance WS2-based photodiodes and transistors used in practical semiconductor applications.
-
Citations
Citations to this article as recorded by- Ultrafast Synthesis of Molybdenum Disulfide via Flashlamp Annealing
Chan Hyeon Yang, Jaehak Lee, Jung Hwan Park
Journal of Powder Materials.2025; 32(6): 509. CrossRef
- Ultrafast Synthesis of Molybdenum Disulfide via Flashlamp Annealing
- [Korean]
- Pressureless Sintering and Microstructure of Pure Tungsten Powders Prepared by Ultrasonic Spray Pyrolysis
- Youn Ji Heo, Eui Seon Lee, Sung-Tag Oh, Jongmin Byun
- J Powder Mater. 2022;29(3):247-251. Published online June 1, 2022
- DOI: https://doi.org/10.4150/KPMI.2022.29.3.247

- 1,613 View
- 11 Download
- 2 Citations
-
 Abstract
Abstract
 PDF
PDF This study demonstrates the effect of the compaction pressure on the microstructure and properties of pressureless-sintered W bodies. W powders are synthesized by ultrasonic spray pyrolysis and hydrogen reduction using ammonium metatungstate hydrate as a precursor. Microstructural investigation reveals that a spherical powder in the form of agglomerated nanosized W particles is successfully synthesized. The W powder synthesized by ultrasonic spray pyrolysis exhibits a relative density of approximately 94% regardless of the compaction pressure, whereas the commercial powder exhibits a relative density of 64% under the same sintering conditions. This change in the relative density of the sintered compact can be explained by the difference in the sizes of the raw powder and the densities of the compacted green body. The grain size increases as the compaction pressure increases, and the sintered compact uniaxially pressed to 50 MPa and then isostatically pressed to 300 MPa exhibits a size of 0.71 m. The Vickers hardness of the sintered W exhibits a high value of 4.7 GPa, mainly due to grain refinement.
-
Citations
Citations to this article as recorded by- Preparation of W-Ni-Cu Alloy Powder by Hydrogen Reduction of Metal Oxides
Youn Ji Heo, Eui Seon Lee, Ji Won Choi, Jongmin Byun, Sung-Tag Oh
Korean Journal of Metals and Materials.2024; 62(5): 334. CrossRef - Influence of the initial powder characteristic on the densified tungsten microstructure by spark plasma sintering and hot isostatic pressing
Ji Young Kim, Eui Seon Lee, Youn Ji Heo, Young-In Lee, Jongmin Byun, Sung-Tag Oh
Powder Metallurgy.2023; 66(5): 644. CrossRef
- Preparation of W-Ni-Cu Alloy Powder by Hydrogen Reduction of Metal Oxides
- [Korean]
- Recycling of Hardmetal Tool through Alkali Leaching Process and Fabrication Process of Nano-sized Tungsten Carbide Powder using Self-propagation High-temperature Synthesis
- Hee-Nam Kang, Dong Il Jeong, Young Il Kim, In Yeong Kim, Sang Cheol Park, Cheol Woo Nam, Seok-Jun Seo, Jin Yeong Lee, Bin Lee
- J Powder Mater. 2022;29(1):47-55. Published online February 1, 2022
- DOI: https://doi.org/10.4150/KPMI.2022.29.1.47

- 1,793 View
- 19 Download
-
 Abstract
Abstract
 PDF
PDF Tungsten carbide is widely used in carbide tools. However, its production process generates a significant number of end-of-life products and by-products. Therefore, it is necessary to develop efficient recycling methods and investigate the remanufacturing of tungsten carbide using recycled materials. Herein, we have recovered 99.9% of the tungsten in cemented carbide hard scrap as tungsten oxide via an alkali leaching process. Subsequently, using the recovered tungsten oxide as a starting material, tungsten carbide has been produced by employing a self-propagating high-temperature synthesis (SHS) method. SHS is advantageous as it reduces the reaction time and is energy-efficient. Tungsten carbide with a carbon content of 6.18 wt % and a particle size of 116 nm has been successfully synthesized by optimizing the SHS process parameters, pulverization, and mixing. In this study, a series of processes for the highefficiency recycling and quality improvement of tungsten-based materials have been developed.
- [Korean]
- Current Status of Smelting and Recycling Technologies of Tungsten
- Ho-Sang Sohn
- J Korean Powder Metall Inst. 2021;28(4):342-351. Published online August 1, 2021
- DOI: https://doi.org/10.4150/KPMI.2021.28.4.342

- 2,308 View
- 58 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF Because of its unique properties, tungsten is a strategic and rare metal used in various industrial applications. However, the world's annual production of tungsten is only 84000 t. Ammonium paratungstate (APT), which is used as the main intermediate in industrial tungsten production, is usually obtained from tungsten concentrates of wolframite and scheelite by hydrometallurgical treatment. Intermediates such as tungsten trioxide, tungsten blue oxide, tungstic acid, and ammonium metatungstate can be derived from APT by thermal decomposition or chemical attack. Tungsten metal powder is produced through the hydrogen reduction of high-purity tungsten oxides, and tungsten carbide powder is produced by the reaction of tungsten powder and carbon black powder at 1300–1700°C in a hydrogen atmosphere. Tungsten scrap can be divided into hard and soft scrap based on shape (bulk or powder). It can also be divided into new scrap generated during the production of tungsten-bearing goods and old scrap collected at the end of life. Recycling technologies for tungsten can be divided into four main groups: direct, chemical, and semi-direct recycling, and melting metallurgy. In this review, the current status of tungsten smelting and recycling technologies is discussed.
-
Citations
Citations to this article as recorded by- Synthesis and thermal properties of single-phase AMT crystals via alcoholic precipitation
Haoyu Liu, Wendi Zhang, Qiusheng Zhou, Xiaobin Li, Jie Li, Guihua Liu, Zhihong Peng, Tiangui Qi, Leiting Shen, Yilin Wang
Canadian Metallurgical Quarterly.2026; 65(2): 2088. CrossRef - The Current Status and Securing Strategies of Core Mineral Tungsten Resources
Dohyun Jeong, Seongmin Kim, Hoseok Jeon
Journal of the Korean Society of Mineral and Energy Resources Engineers.2023; 60(5): 341. CrossRef - Tungsten distribution and vertical migration in soils near a typical abandoned tungsten smelter
Huihui Du, Yang Li, Dan Wan, Chuanqiang Sun, Jing Sun
Journal of Hazardous Materials.2022; 429: 128292. CrossRef
- Synthesis and thermal properties of single-phase AMT crystals via alcoholic precipitation
- [Korean]
- Sintering Behavior and Microstructures of Tantalum and Tantalum-Tungsten Alloys Powders
- Youngmoo Kim, Sung Ho Yang, Seong Lee, Sung Ho Lee, Joon-Woong Noh
- J Korean Powder Metall Inst. 2020;27(5):373-380. Published online October 1, 2020
- DOI: https://doi.org/10.4150/KPMI.2020.27.5.373
- 1,411 View
- 12 Download
- 2 Citations
-
 Abstract
Abstract
 PDF
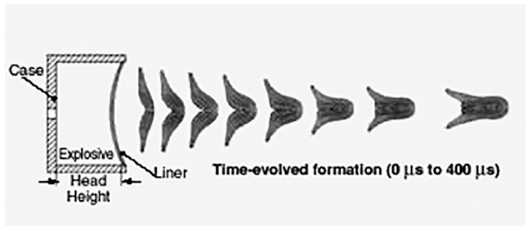
PDF The purpose of this study is to investigate the densification behavior and the corresponding microstructural evolution of tantalum and tantalum-tungsten alloy powders for explosively formed liners. The inherent inhomogeneous microstructures of tantalum manufactured by an ingot metallurgy might degrade the capability of the warhead. Therefore, to overcome such drawbacks, powder metallurgy was incorporated into the near-net shape process in this study. Spark plasma-sintered tantalum and its alloys with finer particle sizes exhibited higher densities and lower grain sizes. However, they were contaminated from the graphite mold during sintering. Higher compaction pressures in die and isostatic compaction techniques also enhanced the sinterability of the tantalum powders; however, a full densification could not be achieved. On the other hand, the powders exhibited full densification after being subjected to hot isostatic pressing over two times. Consequently, it was found that the hot isostatic-pressed tantalum might exhibit a lower grain size and a higher density as compared to those obtained in previous studies.
-
Citations
Citations to this article as recorded by- Understanding the phase evolution and elemental distribution in MoWTaNbVTix manufactured via powder metallurgical approach
Surya T. Bijjala, Ryan Wilkerson, Chad Beamer, Pankaj Kumar
The International Journal of Advanced Manufacturing Technology.2024; 135(11-12): 5925. CrossRef - Thermal Stability and Weight Reduction of Al0.75V2.82CrZr Refractory High Entropy Alloy Prepared Via Mechanical Alloying
Minsu Kim, Hansung Lee, Byungmin Ahn
journal of Korean Powder Metallurgy Institute.2023; 30(6): 478. CrossRef
- Understanding the phase evolution and elemental distribution in MoWTaNbVTix manufactured via powder metallurgical approach
- [Korean]
- Synthesis of W2C by Spark Plasma Sintering of W-WC Powder Mixture and Its Etching Property
- Gyu-Sang Oh, Sung-Min Lee, Sung-Soo Ryu
- J Korean Powder Metall Inst. 2020;27(4):293-299. Published online August 1, 2020
- DOI: https://doi.org/10.4150/KPMI.2020.27.4.293
- 1,131 View
- 10 Download
-
 Abstract
Abstract
 PDF
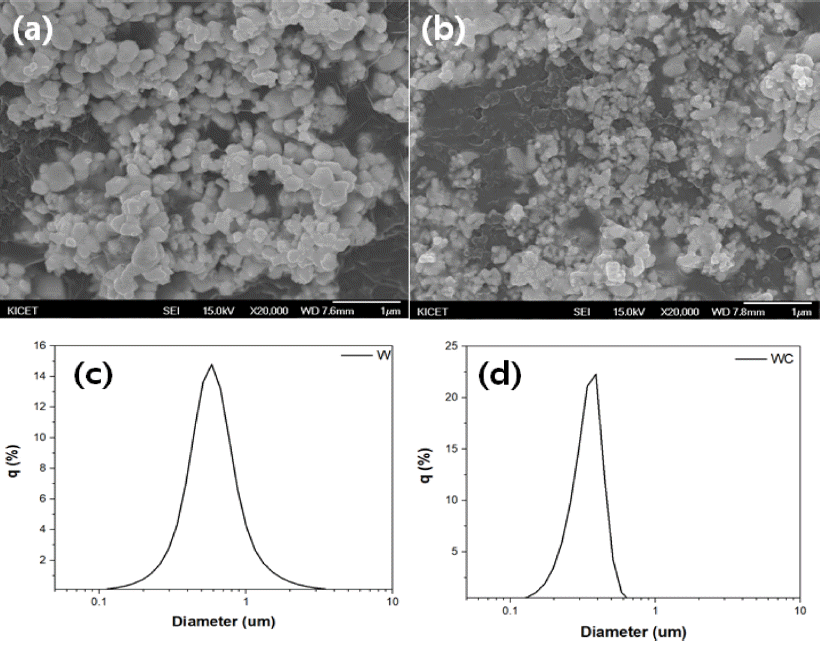
PDF W2C is synthesized through a reaction-sintering process from an ultrafine-W and WC powder mixture using spark plasma sintering (SPS). The effect of various parameters, such as W:WC molar ratio, sintering temperature, and sintering time, on the synthesis behavior of W2C is investigated through X-ray diffraction (XRD) analysis, scanning electron microscopy (SEM) analysis of the microstructure, and final sintered density. Further, the etching properties of a W2C specimen are analyzed. A W2C sintered specimen with a particle size of 2.0 μm and a relative density over 98% could be obtained from a W-WC powder mixture with 55 mol%, after SPS at 1700°C for 20 min under a pressure of 50 MPa. The sample etching rate is similar to that of SiC. Based on X-ray photoelectron spectroscopy (XPS) analysis, it is confirmed that fluorocarbon-based layers such as C-F and C-F2 with lower etch rates are also formed.
- [Korean]
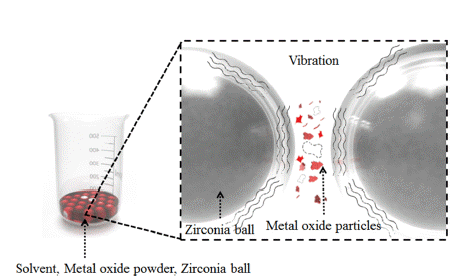
- Investigation on Size Distribution of Tungsten-based Alloy Particles with Solvent Viscosity During Ultrasonic Ball Milling Process
- KeunHyuk Ryu, HyeongSub So, JiSeok Yun, InHo Kim, Kun-Jae Lee
- J Korean Powder Metall Inst. 2019;26(3):201-207. Published online June 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.3.201
- 1,340 View
- 5 Download
- 3 Citations
-
 Abstract
Abstract
 PDF
PDF Tungsten heavy alloys (W–Ni–Fe) play an important role in various industries because of their excellent mechanical properties, such as the excellent hardness of tungsten, low thermal expansion, corrosion resistance of nickel, and ductility of iron. In tungsten heavy alloys, tungsten nanoparticles allow the relatively low-temperature molding of high-melting-point tungsten and can improve densification. In this study, to improve the densification of tungsten heavy alloy, nanoparticles are manufactured by ultrasonic milling of metal oxide. The physical properties of the metal oxide and the solvent viscosity are selected as the main parameters. When the density is low and the Mohs hardness is high, the particle size distribution is relatively high. When the density is high and the Mohs hardness is low, the particle size distribution is relatively low. Additionally, the average particle size tends to decrease with increasing viscosity. Metal oxides prepared by ultrasonic milling in high-viscosity solvent show an average particle size of less than 300 nm based on the dynamic light scattering and scanning electron microscopy analysis. The effects of the physical properties of the metal oxide and the solvent viscosity on the pulverization are analyzed experimentally.
-
Citations
Citations to this article as recorded by- Morphological Control and Surface Modification Characteristics of Nickel Oxalate Synthesized via Oxalic Acid Precipitation
Eunbi Park, Jongwon Bae, Sera Kang, Minsu Kang, Suseong Lee, Kun-Jae Lee
Journal of Powder Materials.2025; 32(5): 375. CrossRef - Manufacture of high sensitive Ag-Fe3O4-PDMS nanocomposite pressure sensor through morphology control of conductive filler
Keunhyuk Ryu, Namhun Kwon, Kun-Jae Lee
Advanced Powder Technology.2021; 32(7): 2441. CrossRef - Grinding behavior of WO3, NiO, Fe2O3 by ultrasonic milling parameters control and preparation of nanocomposite powder
Keunhyuk Ryu, Kun-Jae Lee
Advanced Powder Technology.2020; 31(9): 3867. CrossRef
- Morphological Control and Surface Modification Characteristics of Nickel Oxalate Synthesized via Oxalic Acid Precipitation
- [Korean]
- Fabrication and Characterization of Hexagonal Tungsten Oxide Nanopowders for High Performance Gas Sensing Application
- Jinsoo Park
- J Korean Powder Metall Inst. 2019;26(1):28-33. Published online February 1, 2019
- DOI: https://doi.org/10.4150/KPMI.2019.26.1.28

- 1,148 View
- 6 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF The gas sensor is essential to monitoring dangerous gases in our environment. Metal oxide (MO) gas sensors are primarily utilized for flammable, toxic and organic gases and O3 because of their high sensitivity, high response and high stability. Tungsten oxides (WO3) have versatile applications, particularly for gas sensor applications because of the wide bandgap and stability of WO3. Nanosize WO3 are synthesized using the hydrothermal method. Asprepared WO3 nanopowders are in the form of nanorods and nanorulers. The crystal structure is hexagonal tungsten bronze (MxWO3, x =< 0.33), characterized as a tunnel structure that accommodates alkali ions and the phase stabilizer. A gas detection test reveals that WO3 can detect acetone, butanol, ethanol, and gasoline. This is the first study to report this capability of WO3.
-
Citations
Citations to this article as recorded by- Preparation of potassium tungsten bronze (KxWO3) for near-infrared shielding application via high-energy ball milling: Effect of milling time
Phonlawee Pinthong, Thanaphon Kansaard, Maneerat Songpanit, C.K. Jayasankar, Wanichaya Mekprasart, Kanokthip Boonyarattanakalin, Wisanu Pecharapa
Physica B: Condensed Matter.2026; 734: 418622. CrossRef
- Preparation of potassium tungsten bronze (KxWO3) for near-infrared shielding application via high-energy ball milling: Effect of milling time
- [Korean]
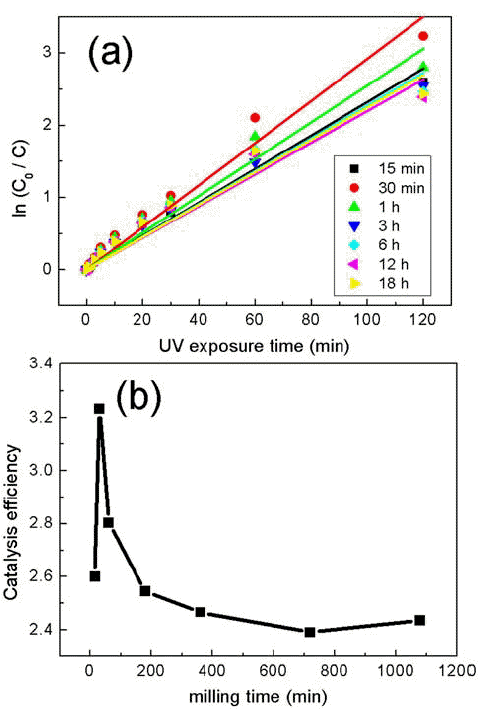
- Photocatalysis of TiO2/WO3 Composites Synthesized by Ball Milling
- Su-Yeol Yu, Chunghee Nam
- J Korean Powder Metall Inst. 2018;25(4):316-321. Published online August 1, 2018
- DOI: https://doi.org/10.4150/KPMI.2018.25.4.316
- 954 View
- 4 Download
-
 Abstract
Abstract
 PDF
PDF Composites of P25 TiO2 and hexagonal WO3 nanorods are synthesized through ball-milling in order to study photocatalytic properties. Various composites of TiO2/WO3 are prepared by controlling the weight percentages (wt%) of WO3, in the range of 1–30 wt%, and milling time to investigate the effects of the composition ratio on the photocatalytic properties. Scanning electron microscopy, x-ray diffraction, and transmission electron microscopy are performed to characterize the structure, shape and size of the synthesized composites of TiO2/WO3. Methylene blue is used as a test dye to analyze the photocatalytic properties of the synthesized composite material. The photocatalytic activity shows that the decomposition efficiency of the dye due to the photocatalytic effect is the highest in the TiO2/WO3 (3 wt%) composite, and the catalytic efficiency decreases sharply when the amount of WO3 is further increased. As the amount of WO3 added increases, dye-removal by adsorption occurs during centrifugation, instead of the decomposition of dyes by photocatalysts. Finally, TiO2/WO3 (3 wt%) composites are synthesized with various milling times. Experimental results show that the milling time has the best catalytic efficiency at 30 min, after which it gradually decreases. There is no significant change after 1 hour.
- [Korean]
- Fabrication of WC/Co composite powder from oxide of WC/Co hardmetal scrap by carbothermal reduction process
- Gil-Geun Lee, Young Soo Lim
- J Korean Powder Metall Inst. 2018;25(3):240-245. Published online June 1, 2018
- DOI: https://doi.org/10.4150/KPMI.2018.25.3.240

- 980 View
- 5 Download
-
 Abstract
Abstract
 PDF
PDF This study focuses on the fabrication of a WC/Co composite powder from the oxide of WC/Co hardmetal scrap using solid carbon in a hydrogen gas atmosphere for the recycling of WC/Co hardmetal. Mixed powders are manufactured by mechanically milling the oxide powder of WC-13 wt% Co hardmetal scrap and carbon black with varying powder/ball weight ratios. The oxide powder of WC-13 wt% Co hardmetal scrap consists of WO3 and CoWO4. The mixed powder mechanically milled at a lower powder/ball weight ratio (high mechanical milling energy) has a more rapid carbothermal reduction reaction in the formation of WC and Co phases compared with that mechanically milled at a higher powder/ball weight ratio (lower mechanical milling energy). The WC/Co composite powder is fabricated at 900°C for 6 h from the oxide of WC/Co hardmetal scrap using solid carbon in a hydrogen gas atmosphere. The fabricated WC/Co composite powder has a particle size of approximately 0.25-0.5 μm.
- [Korean]
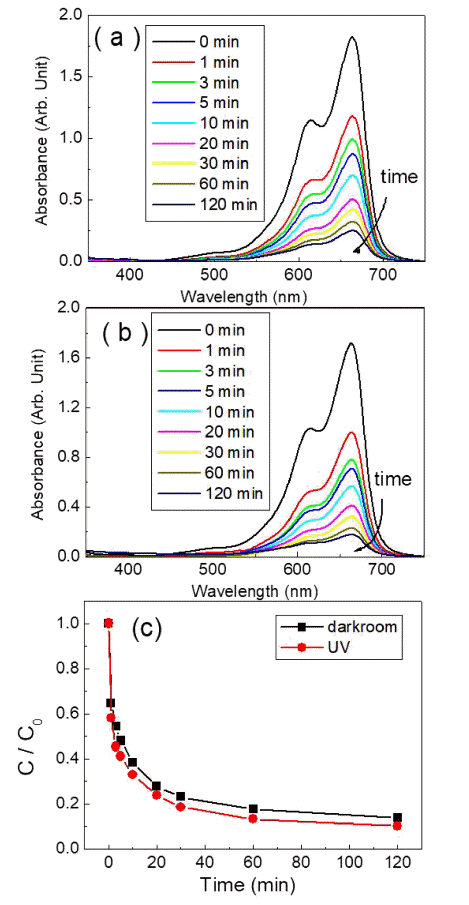
- Photocatalytic and Adsorption Properties of WO3 Nanorods Prepared by Hydrothermal Synthesis
- Su-Yeol Yu, Chunghee Nam
- J Korean Powder Metall Inst. 2017;24(6):483-488. Published online December 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.6.483
- 1,439 View
- 5 Download
- 2 Citations
-
 Abstract
Abstract
 PDF
PDF Transition-metal oxide semiconductors have various band gaps. Therefore, many studies have been conducted in various application fields. Among these, methods for the adsorption of organic dyes and utilization of photocatalytic properties have been developed using various metal oxides. In this study, the adsorption and photocatalytic effects of WO3 nanomaterials prepared by hydrothermal synthesis are investigated, with citric acid added in the hydrothermal process as a structure-directing agent. The nanostructures of WO3 are studied using transmission electron microscopy and scanning electron microscopy images. The crystal structure is investigated using X-ray diffraction patterns, and the changes in the dye concentrations adsorbed on WO3 nanorods are measured with a UV-visible absorption spectrophotometer based on Beer-Lambert’s law. The methylene blue (MB) dye solution is subjected to acid or base conditions to monitor the change in the maximum adsorption amount in relation to the pH. The maximum adsorption capacity is observed at pH 3. In addition to the dye adsorption, UV irradiation is carried out to investigate the decomposition of the MB dye as a result of photocatalytic effects. Significant photocatalytic properties are observed and compared with the adsorption effects for dye removal.
-
Citations
Citations to this article as recorded by- Photocatalytic Properties of WO3 Thin Films Prepared by Electrodeposition Method
Kwang-Mo Kang, Ji-Hye Jeong, Ga-In Lee, Jae-Min Im, Hyun-Jeong Cheon, Deok-Hyeon Kim, Yoon-Chae Nah
Journal of Korean Powder Metallurgy Institute.2019; 26(1): 40. CrossRef - Photocatalysis of TiO<sub>2</sub>/WO<sub>3</sub> Composites Synthesized by Ball Milling
Su-Yeol Yu, Chunghee Nam
Journal of Korean Powder Metallurgy Institute.2018; 25(4): 316. CrossRef
- Photocatalytic Properties of WO3 Thin Films Prepared by Electrodeposition Method
- [Korean]
- Basic Study on the Recycling of Waste Tungsten Scraps by the Oxidation and Reduction Process
- Sang-Uk Kim, Ji-seok Yun, Tae-Wook Kim, Bong-Hwi Cho, In-Ho Kim, Sang-Mu Kim, Chang-Bin Song
- J Korean Powder Metall Inst. 2017;24(1):34-40. Published online February 1, 2017
- DOI: https://doi.org/10.4150/KPMI.2017.24.1.34

- 1,016 View
- 4 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF This study is carried out to obtain basic data regarding oxidation and reduction reactions, originated on the recycling of waste tungsten hard scraps by oxidation and reduction processes. First, it is estimated that the theoretical Gibbs free energy for the formation reaction of WO2 and WO3 are calculated as ΔG1,000K= -407.335 kJ/mol and ΔG1,000K = -585.679 kJ/mol, from the thermodynamics data reported by Ihsan Barin. In the experiments, the oxidation of pure tungsten rod by oxygen is carried out over a temperature range of 700-1,000°C for 1 h, and it is possible to conclude that the oxidation reaction can be represented by a relatively linear relationship. Second, the reduction of WO2 and WO3 powder by hydrogen is also calculated from the same thermodynamics data, and it can be found that it was difficult for the reduction reaction to occur at 1,027°C, in the case of WO2, but it can happen for temperatures higher than 1127°C. On the other hand, WO3 reduction reaction occurs at the relatively low temperature of 827oC. Based on these results, the reduction experiments are carried out at a temperature range of 500-1,000°C for 15 min to 4 h, in the case of WO3 powder, and it is possible to conclude that the reduction at 900°C for 2h is needed for a perfect reduction reaction.
-
Citations
Citations to this article as recorded by- A Basic Study on the Recycling of Wasted Cemented Carbide by the Zn Bath Process(Ⅰ)
Kyung-Sik Kim, In-Ho Kim, Chan-Gi Lee, Chang-Bin Song
Journal of the Korean Institute of Resources Recycling.2020; 29(6): 35. CrossRef
- A Basic Study on the Recycling of Wasted Cemented Carbide by the Zn Bath Process(Ⅰ)
- [English]
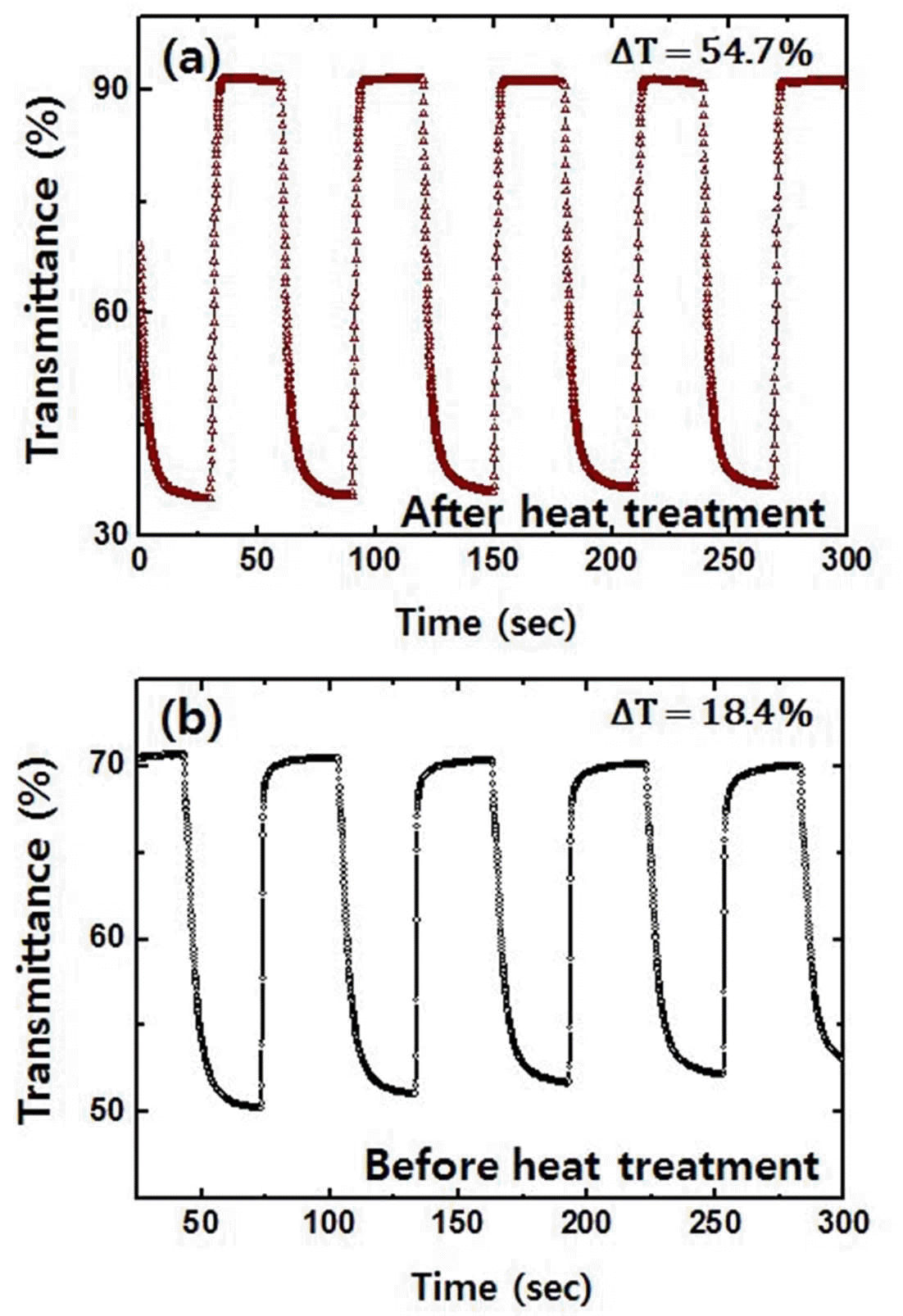
- Synthesis and Characterization of Tungsten Trioxide Films Prepared by a Sol-Gel Method for Electrochromic Applications
- Tae-Ho Kim, Yoon-Chae Nah
- J Korean Powder Metall Inst. 2015;22(5):309-314. Published online October 1, 2015
- DOI: https://doi.org/10.4150/KPMI.2015.22.5.309
- 1,216 View
- 5 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
PDF Tungsten trioxide thin films are successfully synthesized by a sol-gel method using tungsten hexachloride as precursors. The structural, chemical, and optical properties of the prepared films are characterized by scanning electron microscopy, X-ray diffraction, X-ray photoelectron spectroscopy, and UV-Vis spectrophotometry. The electrochemical and electrochromic properties of the films before and after heat treatment are also investigated by cyclic voltammetry, chronoamperometry, and in situ transmittance measurement system. Compared to as-prepared films, heattreated tungsten trioxide thin films exhibit a higher electrochemical reversibility of 0.81 and superior coloration efficiency of 65.7 cm2/C, which implies that heat treatment at an appropriate temperature is a crucial process in a sol-gel method for having a better electrochromic performance.
-
Citations
Citations to this article as recorded by- Temperature-dependent electrochromic cycling performance of solution-processed WO3 films
Hoon Kang, Gyung Hyun Kim, Sungho Kang, Tae Hoon Park, Kwanchul Kim, Hwan Kyu Kim, Yekyung Kim
Nanoscale.2025; 17(35): 20135. CrossRef
- Temperature-dependent electrochromic cycling performance of solution-processed WO3 films
- [Korean]
- Investigation for Microstructure and Hardness of Welded Zone of Cu-Ni Alloy using W92-Ni-Fe Sintering Tool
- Tae-Jin Yoon, Sang-Won Park, Myung-Chang Kang, Joong-Suk Noh, Sung-Wook Chung, Chung-Yun Kang
- J Korean Powder Metall Inst. 2015;22(3):181-186. Published online June 1, 2015
- DOI: https://doi.org/10.4150/KPMI.2015.22.3.181
- 915 View
- 2 Download
-
 Abstract
Abstract
 PDF
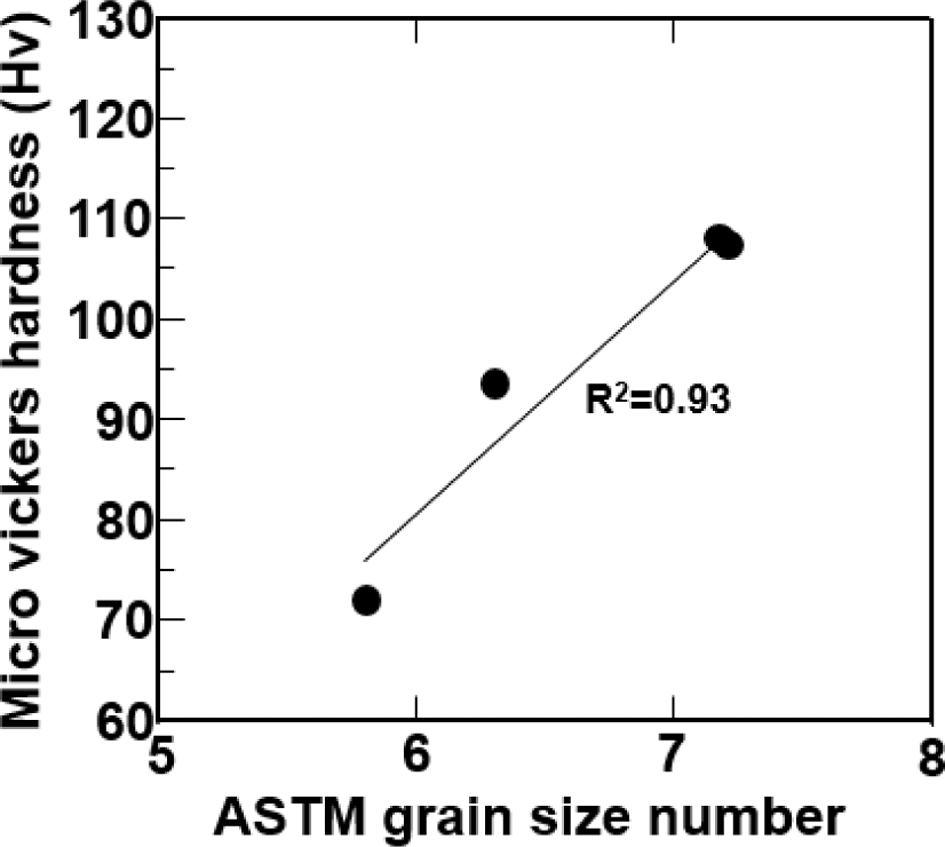
PDF In this study, the effect of the friction stir welding (FSW) was compared with that of the gas tungsten arc welding (GTAW) on the microstructure and microhardness of Cu-Ni alloy weldment. The weldment of 10 mm thickness was fabricated by FSW and GTAW, respectively. Both weldments were compared with each other by optical microstructure, microhardness test and grain size measurement. Results of this study suggest that the microhardness decreased from the base metal (BM) to the heat affected zone (HAZ) and increased at fusion zone (FZ) of GTAW and stir zone (SZ) of FSW. the minimum Hv value of both weldment was obtained at HAZ, respectively, which represents the softening zone, whereas Hv value of FSW weldment was little higher than that of GTAW weldment. These phenomena can be explained by the grain size difference between HAZs of each weldment. Grain size was increased at the HAZ during FSW and GTAW. Because FSW is a solid-state joining process obtaining the lower heat-input generated by rotating shoulder than heat generated in the arc of GTAW.
- [Korean]
- Fabrication of WS2-W-WC Embedded Carbon Nanofiber Composites for Supercapacitors
- Yu-Jin Lee, Hyo-Jin Ahn
- J Korean Powder Metall Inst. 2015;22(2):116-121. Published online April 1, 2015
- DOI: https://doi.org/10.4150/KPMI.2015.22.2.116
- 1,035 View
- 2 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
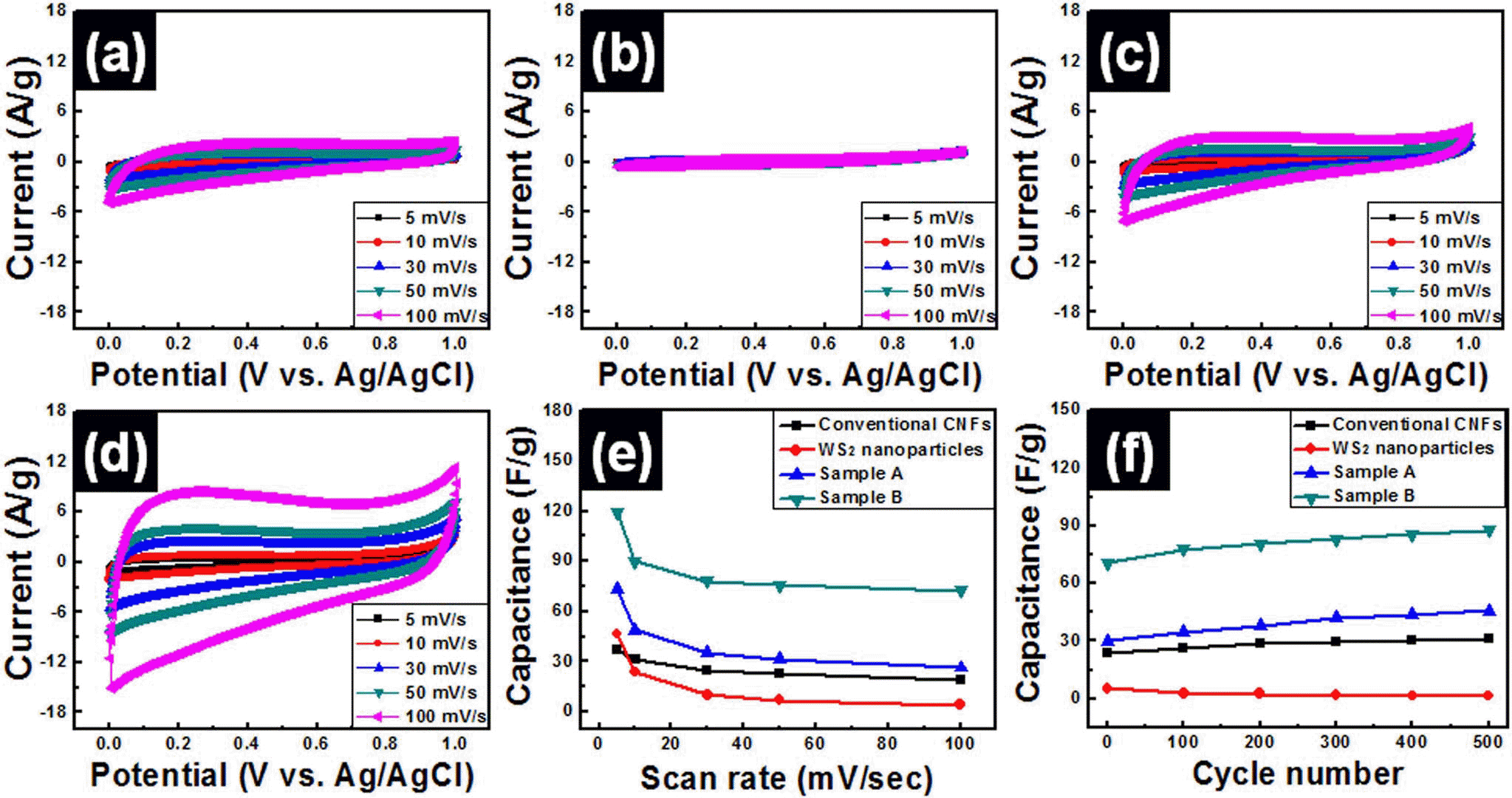
PDF WS2-W-WC embedded carbon nanofiber composites were fabricated by using electrospinning method for use in high-performance supercapacitors. In order to obtain optimum electrochemical properties for supercapacitors, WS2 nanoparticles were used as precursors and the amounts of WS2 precursors were controlled to 4 wt% (sample A) and 8 wt% (sample B). The morphological, structural, and chemical properties of all samples were investigated by means of field emission photoelectron spectroscopy, transmission electron microscopy, X-ray diffraction, and X-ray photoelectron spectroscopy. These results demonstrated that the embedded phases of samples A and B were changed from WS2 to WS2-W-WC through carbothermal reaction during carbonization process. In particular, sample B presented high specific capacitance (~119.7 F/g at 5 mV/s), good high-rate capacitance (~60.5%), and superb cycleability. The enhanced electrochemical properties of sample B were explained by the synergistic effect of the using 1-D structure supports, increase of specific surface area, and improved conductivity from formation of W and WC phases.
-
Citations
Citations to this article as recorded by- WS2 Nanoparticles Embedded in Carbon Nanofibers for a Pseudocapacitor
Ki-Wook Sung, Jung Soo Lee, Tae-Kum Lee, Hyo-Jin Ahn
Korean Journal of Materials Research.2021; 31(8): 458. CrossRef
- WS2 Nanoparticles Embedded in Carbon Nanofibers for a Pseudocapacitor
- [Korean]
- Effect of Oxygen Content in the Tungsten Powder Fabricated by Electrical Explosion of Wire Method on the Behavior of Spark-Plasma Sintering
- Cheol-Hee Kim, Seong Lee, Byung-Kee Kim, Ji Soon Kim
- J Korean Powder Metall Inst. 2014;21(6):447-453. Published online December 1, 2014
- DOI: https://doi.org/10.4150/KPMI.2014.21.6.447
- 848 View
- 4 Download
- 1 Citations
-
 Abstract
Abstract
 PDF
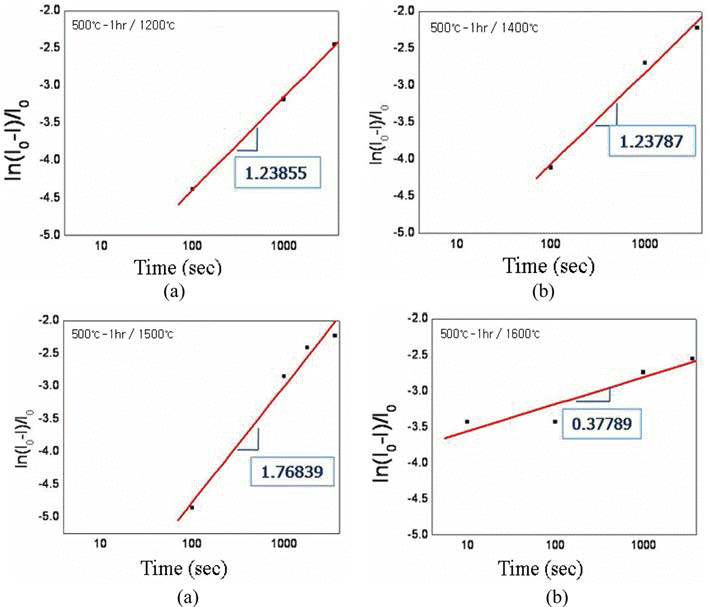
PDF Effect of oxygen content in the ultrafine tungsten powder fabricated by electrical explosion of wire method on the behvior of spark plasma sintering was investigated. The initial oxygen content of 6.5 wt% of as-fabricated tungsten powder was reduced to 2.3 and 0.7 wt% for the powders which were reduction-treated at 400°C for 2 hour and at 500°C for 1h in hydrogen atmosphere, respectively. The reduction-treated tungsten powders were spark-plasma sintered at 1200-1600°C for 100-3600 sec. with applied pressure of 50 MPa under vacuum of 0.133 Pa. Maximun sindered density of 97% relative density was obtained under the condition of 1600°C for 1h from the tungsten powder with 0.7 wt% oxygen. Sintering activation energy of 95.85 kJ/mol−1 was obtained, which is remarkably smaller than the reported ones of 380~460 kJ/mol−1 for pressureless sintering of micron-scale tungsten powders.
-
Citations
Citations to this article as recorded by- Effect of Powder Mixing Process on the Characteristics of Hybrid Structure Tungsten Powders with Nano-Micro Size
Na-Yeon Kwon, Young-Keun Jeong, Sung-Tag Oh
Journal of Korean Powder Metallurgy Institute.2017; 24(5): 384. CrossRef
- Effect of Powder Mixing Process on the Characteristics of Hybrid Structure Tungsten Powders with Nano-Micro Size
TOP
 KPMI
KPMI


 First
First Prev
Prev


